Crossing the Barrier: Molecular Weight, Lipophilicity, and BBB Permeability in CNS Drug Development
This article provides a comprehensive analysis of the two primary physicochemical determinants of Blood-Brain Barrier (BBB) permeability: molecular weight (MW) and lipophilicity (often measured as LogP/LogD).
Crossing the Barrier: Molecular Weight, Lipophilicity, and BBB Permeability in CNS Drug Development
Abstract
This article provides a comprehensive analysis of the two primary physicochemical determinants of Blood-Brain Barrier (BBB) permeability: molecular weight (MW) and lipophilicity (often measured as LogP/LogD). Targeting researchers, scientists, and drug development professionals, it explores the foundational science of these factors, details methodological approaches for prediction and measurement, discusses troubleshooting strategies for suboptimal compounds, and evaluates validation techniques and comparative frameworks. The review synthesizes current understanding with recent advancements to guide the rational design of CNS-active therapeutics.
The Physical Chemistry of Brain Access: Why MW and LogP Rule the BBB
The blood-brain barrier (BBB) is a highly specialized, selective interface between the peripheral circulation and the central nervous system (CNS). Its primary function is to maintain cerebral homeostasis and protect the neural microenvironment from toxins and pathogens. For neurotherapeutics, the BBB represents the single most significant obstacle to drug delivery. Understanding the physicochemical determinants of BBB permeability—specifically molecular weight and lipophilicity—is therefore a cornerstone of modern CNS drug development research.
Core Physicochemical Determinants of BBB Permeability
The passive diffusion of molecules across the BBB endothelial cell membrane is principally governed by a limited set of physicochemical properties. These factors are central to predictive modeling and rational drug design.
Table 1: Key Physicochemical Factors Influencing Passive BBB Permeability
| Factor | Optimal Range for Passive Diffusion | Rationale & Impact |
|---|---|---|
| Molecular Weight (MW) | Typically <400-500 Da | Smaller size facilitates transcellular diffusion through the lipid bilayer. |
| Lipophilicity | Log P (octanol/water) ~1.5-3.0 | Adequate lipid solubility is required for membrane partitioning. Excessive lipophilicity can increase plasma protein binding and clearance. |
| Hydrogen Bond Donors (HBD) | <3 | Polar HBD groups decrease membrane permeability by increasing desolvation energy. |
| Hydrogen Bond Acceptors (HBA) | <8 | Similar to HBDs, excessive HBA count reduces lipophilicity and impedes diffusion. |
| Polar Surface Area (PSA) | <60-90 Ų | A lower PSA correlates with reduced hydrogen bonding potential and higher permeability. |
Experimental Protocols for Assessing BBB Permeability
In VitroBBB Model Using hCMEC/D3 Cell Monolayers
This protocol details the use of a human immortalized brain endothelial cell line to model BBB permeability in vitro.
Protocol:
- Cell Culture: Seed hCMEC/D3 cells (passage 25-35) onto rat tail collagen-I coated Transwell inserts (0.4 µm pore, 12-well format) at a density of 1.2 x 10^5 cells/cm².
- Differentiation: Culture cells for 5-7 days in EGM-2 MV medium supplemented with 1% fetal bovine serum (FBS), 1.4 µM hydrocortisone, 5 µg/mL ascorbic acid, and 1 ng/mL basic fibroblast growth factor (bFGF). Change medium every 48 hours.
- Integrity Validation: Measure Transendothelial Electrical Resistance (TEER) daily using a volt-ohm meter. Accept monolayers with TEER >40 Ω·cm². Perform a sodium fluorescein (376 Da) permeability assay post-experiment; apparent permeability (Papp) should be < 2.0 x 10^-6 cm/s.
- Permeability Assay: Dilute test compound in pre-warmed Hanks' Balanced Salt Solution (HBSS) with 10 mM HEPES (pH 7.4). Add to the donor compartment (apical for A→B, basolateral for B→A). Sample from the receiver compartment at e.g., 30, 60, 90, and 120 minutes.
- Quantification: Analyze samples using LC-MS/MS. Calculate Papp (cm/s):
Papp = (dQ/dt) / (A * C0), where dQ/dt is the flux rate, A is the membrane area, and C0 is the initial donor concentration.
In SituBrain Perfusion in Rodents
This gold-standard in vivo technique provides direct measurement of unidirectional brain uptake clearance.
Protocol:
- Surgical Preparation: Anesthetize a rat (e.g., Sprague-Dawley) with ketamine/xylazine. Cannulate the common carotid artery.
- Perfusion: Connect the cannula to a perfusion pump delivering oxygenated, warmed (37°C) Krebs-Ringer bicarbonate buffer (pH 7.4) containing the test compound and a vascular space marker (e.g., [14C]-sucrose).
- Perfusion Phase: Initiate perfusion at a constant flow rate (e.g., 4 mL/min) for a short, defined time (15-120 seconds). Immediately decapitate the animal at the end of perfusion.
- Tissue Processing: Rapidly remove the ipsilateral hemisphere and dissect specific brain regions. Homogenize and digest tissues.
- Analysis: Quantify compound concentration in brain and perfusion fluid via scintillation counting (for radiolabeled compounds) or LC-MS/MS. Calculate the Brain Uptake Clearance (Kin, mL/s/g):
Kin = (C_brain - C_vasc) / (T * C_perfusate), where C_vasc is corrected using the vascular marker.
Visualization of Pathways and Workflows
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for BBB Permeability Research
| Item | Function & Rationale | Example Product/Source |
|---|---|---|
| hCMEC/D3 Cell Line | Immortalized human cerebral microvascular endothelial cells; standard for in vitro BBB models. | Merck Millipore (SCC066) |
| Transwell Permeable Supports | Polyester/Collagen-coated inserts with defined pore size (0.4 µm) for forming cell monolayers. | Corning (Costar) |
| EVOM3 Voltohmmeter | For accurate, daily measurement of Transendothelial Electrical Resistance (TEER). | World Precision Instruments |
| Rat Tail Collagen, Type I | Extracellular matrix coating to support endothelial cell adhesion and differentiation. | Corning (354236) |
| Sodium Fluorescein | Low-MW paracellular flux marker to validate monolayer integrity post-assay. | Thermo Fisher Scientific |
| [14C]-Sucrose / [3H]-Inulin | Radiolabeled vascular space markers for in situ brain perfusion studies. | American Radiolabeled Chemicals |
| LC-MS/MS System | Gold-standard for sensitive and specific quantification of test compounds in biological matrices. | e.g., Waters Xevo TQ-S, Sciex Triple Quad 6500+ |
| Oxygenated Krebs-Ringer Buffer | Physiological perfusion medium for ex vivo and in situ experiments. | Custom-prepared or commercial aCSF. |
The 500 Dalton rule, a heuristic for predicting blood-brain barrier (BBB) permeability, posits that molecules with a molecular weight (MW) below 500 Daltons are more likely to cross the BBB via passive diffusion. This in-depth technical guide examines the biochemical and physiological foundations of this rule within the broader context of BBB permeability factors—molecular weight, lipophilicity, and other key descriptors. We review the historical evidence supporting the rule, detail modern experimental and computational challenges to its universality, and provide updated frameworks for CNS drug development.
The 500 Dalton rule emerged from seminal analyses of known drugs, demonstrating a stark drop in the likelihood of passive BBB penetration as molecular weight increases beyond 500 Da. This threshold is intrinsically linked to other physicochemical properties, primarily lipophilicity (often measured as Log P or Log D), creating a multifactorial permeability profile.
Core Physicochemical Determinants of BBB Permeability
BBB permeability via passive transcellular diffusion is governed by a combination of factors:
- Molecular Weight (MW): Impacts the diffusion coefficient; larger molecules diffuse more slowly.
- Lipophilicity (Log P/Log D): Determines partitioning into the lipid bilayer of endothelial cells. Optimal Log P is often cited between 1.5 and 3.0.
- Polar Surface Area (PSA): A measure of a molecule's hydrogen-bonding potential; PSA > 60–70 Ų typically reduces permeability.
- Hydrogen Bond Donor/Count: High counts impede membrane partitioning.
Table 1: Historical Permeability Guidelines (Rule-of-5 Derivatives)
| Property | Classic "Rule of 5" for Oral Drugs | Typical CNS Drug Optimal Range | Primary Influence on BBB Penetration |
|---|---|---|---|
| Molecular Weight | < 500 Da | < 450 Da | Diffusion rate, paracellular exclusion |
| cLogP | < 5 | 2 – 4 | Lipid bilayer partitioning |
| H-bond Donors | < 5 | < 3 | Desolvation energy for membrane crossing |
| H-bond Acceptors | < 10 | < 7 | Desolvation energy, polar surface area |
| Polar Surface Area | Not specified | < 60-70 Ų | Hydrogen bonding with membrane/water |
Modern Exceptions and Challenges to the 500 Dalton Rule
The rule is a guideline, not a law. Numerous exceptions exist, driven by specific structural and physiological mechanisms.
- High MW Permeants (MW > 500 Da): Certain molecules, such as some macrolides and peptides, can cross via passive diffusion if they are highly lipophilic and flexible, or via active transport.
- Low MW Non-Permeants (MW < 500 Da): Molecules with high polarity, excessive H-bonding, or that are substrates for efflux transporters (e.g., P-glycoprotein) may show negligible brain penetration despite a low MW.
- The Role of Transporters: Carrier-Mediated Transport (CMT) and Receptor-Mediated Transcytosis (RMT) enable the passage of nutrients, hormones, and drug conjugates well above the 500 Da limit.
Table 2: Documented Exceptions to the 500 Dalton Rule
| Compound Class/Example | Approx. MW (Da) | BBB Permeability | Reason for Exception |
|---|---|---|---|
| Cyclosporin A | 1202 | Low (P-gp substrate) | Classic efflux transporter substrate. |
| Some Synthetic Opioids | 450-600 | High | Optimized lipophilicity & low PSA override MW limit. |
| Antibody Fragments | ~25,000 | Very Low (unless via RMT) | Utilize receptor-mediated transcytosis (e.g., transferrin receptor). |
| L-Dopa | 197 | High (for a carboxylate) | Substrate for the large neutral amino acid transporter (LAT1 - CMT). |
| Morphine | 285 | Lower than predicted | Moderate P-gp substrate and H-bond donor count. |
Experimental Protocols for Assessing BBB Permeability
1In VitroBBB Model (Transwell Assay)
Objective: To measure the apparent permeability (Papp) of a compound across a monolayer of brain endothelial cells. Detailed Protocol:
- Cell Culture: Seed immortalized human brain microvascular endothelial cells (hBMECs) on collagen-coated polyester transwell inserts (0.4 µm pore size) at high density.
- Barrier Integrity Validation: Measure Transendothelial Electrical Resistance (TEER) daily using a volt-ohm meter. Accept only monolayers with TEER > 150 Ω·cm². Perform a tracer flux assay (e.g., sodium fluorescein, MW 376 Da) to confirm low paracellular leakage.
- Permeability Assay: Add test compound to the donor compartment (apical for blood-to-brain direction). Sample from the acceptor compartment (basolateral) at e.g., 30, 60, 90, and 120 minutes.
- Analytical Quantification: Use LC-MS/MS to quantify compound concentrations in samples.
- Data Calculation: Calculate Papp (cm/s) using the formula: Papp = (dQ/dt) / (A * C0), where dQ/dt is the flux rate, A is the membrane area, and C0 is the initial donor concentration.
- Efflux Ratio Assessment: Repeat assay with compound in basolateral-to-apical direction. An efflux ratio (Papp(B-A)/Papp(A-B)) > 2 suggests active efflux.
2In SituBrain Perfusion in Rodents
Objective: To determine the unidirectional brain uptake clearance (Kin) without confounding systemic factors. Detailed Protocol:
- Surgical Preparation: Anesthetize rat. Cannulate the common carotid artery.
- Perfusion Solution: Prepare a physiological buffer (e.g., Krebs-bicarbonate) containing the test compound and a vascular space marker (e.g., [14C]-sucrose).
- Perfusion: Start perfusion via the carotid cannula at a constant flow rate (e.g., 2.5 mL/min) for a short, defined time (15-60 seconds). Immediately decapitate the animal at the end of perfusion.
- Tissue Processing: Dissect brain regions (cortex, striatum, etc.). Homogenize and digest tissues.
- Quantification: Use scintillation counting (for radiolabeled compounds) or LC-MS/MS to measure compound in brain and perfusion fluid.
- Data Calculation: Correct brain concentration for vascular entrapment using the sucrose space. Calculate Kin = (Qbrain - Qvascular) / (Perfusion time * Perfusate concentration), where Q is quantity.
Visualization of Key Concepts
Diagram Title: Pathways to BBB Penetration Beyond Passive Diffusion
Diagram Title: Integrated Experimental Workflow for BBB Permeability Assessment
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for BBB Permeability Research
| Item | Function & Application | Example/Supplier Note |
|---|---|---|
| hBMEC/Primary Cells | Provide the biological barrier component for in vitro models. Express key junctions & transporters. | Immortalized hBMEC lines (e.g., hCMEC/D3); primary rat BMECs. |
| Transwell Inserts | Permeable supports for growing cell monolayers, allowing separate access to apical & basolateral compartments. | Polyester membrane, 0.4 µm pore, various diameters (12, 24-well). |
| TEER Measurement System | Voltmeter/electrode system to non-invasively monitor barrier integrity and tight junction formation. | EVOM2 (World Precision Instruments) or cellZscope (nanoAnalytics). |
| LC-MS/MS System | Gold-standard for sensitive and specific quantification of test compounds in complex biological matrices. | Required for in vitro and in vivo sample analysis. |
| P-gp/BCRP Substrates & Inhibitors | Pharmacological tools to identify and characterize active efflux mechanisms. | Substrates: Digoxin, Rhodamine 123. Inhibitors: Elacridar, Ko143. |
| BBB Permeability Kits | Pre-configured assay kits for rapid PAMPA (Parallel Artificial Membrane Permeability Assay) screening. | PAMPA-BBB Kit (e.g., from pION). Provides high-throughput passive permeability rank. |
| Radio/Sterile Perfusion Pumps | Essential for conducting in situ brain perfusion studies with precise flow control. | Syringe pumps allowing mL/min flow rates. |
| Tight Junction Markers | Antibodies for immunofluorescence validation of barrier morphology (e.g., ZO-1, Claudin-5). | Used in conjunction with TEER for model validation. |
The 500 Dalton rule remains a valuable initial filter in CNS drug design. However, modern drug discovery must view it through the lens of a more sophisticated, multifactorial paradigm. Success depends on optimizing the balance of MW, lipophilicity, PSA, and hydrogen-bonding capacity while strategically evading efflux transporters or engaging endogenous influx mechanisms. Future progress hinges on advanced predictive models that integrate in silico, in vitro, and in vivo data, and on novel technologies that safely leverage CMT and RMT pathways to deliver therapeutic agents across the BBB, irrespective of molecular weight.
Within the critical research framework of Blood-Brain Barrier (BBB) permeability, lipophilicity stands as a principal determinant alongside molecular weight and ionization state. The broader thesis posits that successful CNS drug candidates must optimize these factors to harness passive diffusion. This whitepaper provides an in-depth technical analysis of the core lipophilicity parameters—LogP and LogD—detailing their measurement, interpretation, and definitive role in governing passive transmembrane movement, a non-saturable process vital for bioavailability.
Core Definitions and Theoretical Foundation
Lipophilicity is the affinity of a molecule for a lipophilic environment. It is quantitatively described by two key parameters:
- LogP: The partition coefficient (P) is the ratio of the concentrations of a neutral (unionized) compound in a two-phase system, typically 1-octanol and water. LogP is its logarithm (base 10). It is a constant for a given compound at a specified temperature.
- LogD: The distribution coefficient (D) accounts for the ionization state of a molecule at a specific pH. It represents the ratio of the sum of all forms of the compound (ionized and unionized) in octanol to the sum of all forms in water. LogD is pH-dependent.
For passive diffusion, only the neutral, unionized form of a molecule is considered permeable across lipid bilayers. Therefore, LogD provides a more physiologically relevant metric as it reflects the lipophilicity of the mixture of species present at a biological pH (e.g., pH 7.4).
Table 1: Benchmark LogP/LogD Values and BBB Permeability Correlation
| LogP/LogD (at pH 7.4) Range | Interpretation for Passive Diffusion & BBB Penetration |
|---|---|
| < 1 | High hydrophilicity. Poor membrane permeability, likely restricted to paracellular transport. |
| 1 – 3 | Optimal range for most oral drugs and potential CNS activity. Favors balance of solubility and permeability. |
| > 3 – 5 | High lipophilicity. May lead to poor aqueous solubility, increased metabolic clearance, and non-specific binding. |
| > 5 | Excessive lipophilicity. Very poor solubility, high plasma protein binding, and low brain uptake likely. |
Table 2: LogP and LogD (pH 7.4) of Representative Drugs
| Compound | Calculated LogP (cLogP) | Experimental LogD (pH 7.4) | BBB Permeability (Classification) |
|---|---|---|---|
| Caffeine | -0.07 | ~0.0 | High (CNS active) |
| Diazepam | 2.99 | 2.82 | High (CNS active) |
| Warfarin | 2.70 | 1.40 | Low (High plasma protein binding) |
| Propranolol | 3.48 | 1.16 | High (CNS side effects) |
| Ranitidine | 0.27 | -0.3 | Very Low (Permanently charged) |
Experimental Protocols for Determination
Shake-Flask Method (Gold Standard)
Principle: Direct measurement of compound distribution between octanol and aqueous buffer. Detailed Protocol:
- Preparation: Pre-saturate 1-octanol and the aqueous buffer (e.g., phosphate buffer pH 7.4) with each other by shaking equal volumes together for 24 hours, then allowing separation.
- Partitioning: Dissolve the compound in the pre-saturated phase where it is most soluble. Combine this phase with an equal volume of the other pre-saturated phase in a glass vial (typical total volume 2-10 mL).
- Equilibration: Shake the mixture vigorously for 1 hour at constant temperature (e.g., 25°C) to reach partitioning equilibrium.
- Separation: Centrifuge the mixture (e.g., 3000 rpm, 10 min) to achieve complete phase separation.
- Quantification: Carefully separate the two phases. Quantify the concentration of the compound in each phase using a validated analytical method (e.g., HPLC-UV, LC-MS/MS).
- Calculation:
- For LogP (neutral compounds): Use a buffer at a pH where the compound is >99% unionized.
P = [Compound]ₒcₜₐₙₒₗ / [Compound]wₐₜₑᵣLogP = log₁₀(P)- For LogD at a specific pH:
D = [Compound]ₒcₜₐₙₒₗ,ₜₒₜₐₗ / [Compound]wₐₜₑᵣ,ₜₒₜₐₗ LogD = log₁₀(D)
Reversed-Phase HPLC (RP-HPLC) Method
Principle: Correlation between chromatographic retention time and lipophilicity. Detailed Protocol:
- Column: Use a C18 or other octadecyl-silylated stationary phase column.
- Mobile Phase: Utilize an isocratic or gradient elution with a mixture of water and a water-miscible organic solvent (e.g., methanol, acetonitrile).
- Calibration: Run a series of standards with known LogP values (e.g., from the shake-flask method).
- Measurement: Inject the test compound and measure its retention time (k, capacity factor).
- Correlation: Plot log k of the standards against their known LogP to create a linear calibration curve. The LogP of the unknown is extrapolated from this curve.
Visualizing Relationships and Workflows
Diagram 1: The Role of LogP and LogD in BBB Passive Diffusion
Diagram 2: Shake-Flask Experiment Workflow
The Scientist's Toolkit: Key Research Reagents & Materials
Table 3: Essential Reagents for Lipophilicity Measurement
| Item | Function & Specification |
|---|---|
| 1-Octanol (HPLC Grade) | Organic model phase representing lipid membranes. Must be pre-saturated with buffer. |
| Phosphate Buffer Salts (e.g., Na₂HPO₄, KH₂PO₄) | For preparing precise aqueous phases at physiological pH (e.g., 7.4) and other pH values for LogD profiles. |
| HPLC-MS Grade Water & Solvents (MeOH, ACN) | For mobile phase preparation in RP-HPLC methods and sample dilution for analysis. |
| C18 Reversed-Phase HPLC Column | Stationary phase for chromatographic determination of lipophilicity indices (e.g., LogK₍w₎). |
| LogP Standard Kit | A set of compounds with reliably known shake-flask LogP values for calibrating chromatographic or computational methods. |
| pH Meter with ISFET Electrode | Accurate pH measurement of aqueous buffers; ISFET electrodes are compatible with octanol-contaminated samples. |
| Centrifuge & Glass Vials | For rapid, clean separation of octanol/buffer phases post-equilibration. |
| Analytical Balance (0.01 mg sensitivity) | Precise weighing of compound and buffer salts for solution preparation. |
The Lipinski 'Rule of 5' (Ro5) is a foundational heuristic in drug discovery, predicting oral bioavailability based on physicochemical properties. For Central Nervous System (CNS) drugs, the primary challenge is not just absorption but efficient Blood-Brain Barrier (BBB) permeation. This guide examines the critical adaptations of the Ro5 for CNS drug design within the broader context of BBB permeability research, focusing on molecular weight (MW), lipophilicity, and other key factors.
Core Physicochemical Properties for BBB Permeation
BBB permeability is governed by passive diffusion and active transport mechanisms. While the original Ro5 sets thresholds for oral drugs, CNS-specific adaptations emphasize a narrower optimal range.
Table 1: Original Ro5 vs. CNS-Adapted Guidelines
| Property | Original Rule of 5 (Oral Drugs) | CNS-Adapted Target (Optimal) | Rationale for CNS Adaptation |
|---|---|---|---|
| Molecular Weight (MW) | ≤ 500 Da | 350 - 450 Da | Lower MW favors passive diffusion across the tight endothelial junctions of the BBB. |
| Lipophilicity (cLogP) | ≤ 5 | cLogP: 2 - 4cLogD₇.₄: 2 - 3 | Optimal lipophilicity balances membrane permeability and avoidance of non-specific binding, P-gp efflux, and high metabolic clearance. |
| Hydrogen Bond Donors (HBD) | ≤ 5 | ≤ 2 | Reduction in HBD count minimizes desolvation energy and hydrogen-bonding with BBB efflux transporters. |
| Hydrogen Bond Acceptors (HBA) | ≤ 10 | ≤ 6 | Lower HBA count reduces polarity and improves lipophilicity for passive diffusion. |
| Polar Surface Area (PSA) | Not specified | ≤ 70 Ų (ideally < 60 Ų) | Low PSA is a critical predictor of passive transcellular diffusion through lipid membranes. |
Limitations of the Adapted Rules
While these adaptations provide valuable guidance, they present significant limitations:
- Oversimplification of Transport Mechanisms: The rules primarily model passive diffusion, neglecting the critical roles of active influx (e.g., via LAT1, GLUT1) and efflux transporters (e.g., P-glycoprotein).
- Neglect of Molecular Flexibility: Properties like number of rotatable bonds (NRB) and 3D polar surface area can significantly impact permeability but are not captured.
- The "3-Dimensional" Nature of Properties: Lipophilicity (LogP) and acidity/basicity (pKa) are interrelated, determining the distribution coefficient (LogD) at physiological pH (7.4), which is a more accurate predictor.
- Species Differences: BBB transporter expression and function can vary between preclinical species and humans, complicating translation.
Key Experimental Protocols for Assessing BBB Permeability
1. In Vitro BBB Model: hCMEC/D3 Monolayer Assay
- Objective: To predict passive and active transcellular permeability in a human cell-based model.
- Protocol: a. Culture human cerebral microvascular endothelial cells (hCMEC/D3) on collagen-coated Transwell inserts until a tight monolayer forms (TEER > 40 Ω·cm²). b. Dissolve test compound in assay buffer (e.g., Hanks' Balanced Salt Solution, HBSS, pH 7.4). c. Add compound to the donor (apical) compartment. Sample from both donor and acceptor (basolateral) compartments at multiple time points (e.g., 30, 60, 120 min). d. Quantify compound concentration using LC-MS/MS. e. Calculate apparent permeability (Papp): Papp = (dQ/dt) / (A * C₀), where dQ/dt is the transport rate, A is the membrane area, and C₀ is the initial donor concentration. f. Include a P-gp substrate (e.g., Digoxin) and inhibitor (e.g., Zosuquidar) to assess efflux transporter involvement (Efflux Ratio = Papp(B→A)/Papp(A→B)).
2. In Situ Brain Perfusion in Rodents
- Objective: To measure unidirectional brain uptake clearance (Kin) without confounding effects of systemic clearance or plasma protein binding.
- Protocol: a. Anesthetize and cannulate the common carotid artery of a rat or mouse. b. Perfuse a physiologically buffered saline solution containing the test compound and a vascular reference marker (e.g., [¹⁴C]-Sucrose, which does not cross the BBB) for a short, defined time (15-60 seconds). c. Terminate perfusion by decapitation. Rapidly remove and homogenize the ipsilateral hemisphere. d. Quantify drug and reference marker in brain homogenate and perfusate via scintillation counting or LC-MS/MS. e. Calculate Kin: K*in* = (Q*brain - V_vascular) / (T * C*perfusate*)*, where Qbrain is total brain drug, Vvascular is the vascular volume (from reference marker), T is perfusion time, and Cperfusate is drug concentration in perfusate.
Visualizations
Title: Key Pathways for Molecular Crossing of the Blood-Brain Barrier
Title: CNS Drug Property Impact Network
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for BBB Permeability Research
| Item / Reagent | Function / Application |
|---|---|
| hCMEC/D3 Cell Line | Immortalized human cerebral microvascular endothelial cells for constructing physiologically relevant in vitro BBB models. |
| Transwell Permeable Supports | Collagen-coated polycarbonate membrane inserts for growing endothelial cell monolayers and performing permeability assays. |
| LC-MS/MS System | Gold-standard analytical instrument for sensitive and specific quantification of drug concentrations in biological matrices (plasma, brain homogenate). |
| Radioisotopes (e.g., ¹⁴C, ³H) | Used as tracers in in situ brain perfusion and in vivo pharmacokinetic studies to accurately measure uptake and distribution. |
| P-gp Substrates/Inhibitors (e.g., Digoxin, Zosuquidar) | Pharmacological tools to characterize the role of the key efflux transporter P-glycoprotein in limiting brain exposure. |
| TEER Measurement System | Measures Trans-Endothelial Electrical Resistance to validate the integrity and tight junction formation of in vitro BBB monolayers. |
The Lipinski 'Rule of 5' provides a crucial starting point for CNS drug design but requires significant adaptations focusing on lower MW, moderate lipophilicity (cLogD), and reduced hydrogen bonding. These adapted rules are best used as a qualitative guide rather than a strict filter. Successful CNS drug discovery necessitates integrating these guidelines with advanced in vitro and in situ experimental models that account for active transport, and with computational models that leverage multi-parameter optimization to navigate the complex trade-offs between permeability, solubility, and metabolic stability.
In the pursuit of central nervous system (CNS) therapeutics, predicting Blood-Brain Barrier (BBB) permeability remains a paramount challenge. Historically, simplistic rules, such as Lipinski’s Rule of 5 (MW ≤ 500, LogP ≤ 5), served as initial filters. However, CNS drug discovery necessitates more nuanced descriptors. This whitepaper contextualizes BBB permeability within a modern molecular property space, arguing that optimal permeability arises from the complex, non-linear interplay of Molecular Weight (MW), lipophilicity (often measured as LogP or LogD), Polar Surface Area (PSA), and Hydrogen Bond Donor/Acceptor counts (HBD/HBA). Transcending simple thresholds to model their synergistic effects is critical for rational design.
Quantitative Descriptor Landscape for BBB Permeability
The following table summarizes key molecular descriptors, their typical optimal ranges for BBB penetration, and their physiological interpretation.
Table 1: Core Molecular Descriptors Governing BBB Permeability
| Descriptor | Typical Optimal Range for CNS+ | Physiological Rationale | Measurement/Calculation |
|---|---|---|---|
| Molecular Weight (MW) | <450 Da | Reduced passive diffusion; increased efflux likelihood. | Sum of atomic masses. |
| Lipophilicity (LogP/LogD₇.₄) | LogD₇.₄: 1-4 | Balances membrane partitioning (too low) vs. aqueous phase solubility and protein binding (too high). | LogP: Octanol/water partition coeff. LogD: Distribution coeff. at pH 7.4. |
| Polar Surface Area (PSA) | <90 Ų (pref. <70 Ų) | Proxy for desolvation energy; high PSA impedes passive diffusion through lipid bilayer. | Sum of surfaces of polar atoms (O, N, attached H). |
| Hydrogen Bond Donors (HBD) | ≤3 | Form strong H-bonds with water, increasing desolvation cost. | Count of OH and NH groups. |
| Hydrogen Bond Acceptors (HBA) | ≤7 | Similar desolvation penalty as HBDs, but generally less restrictive. | Count of N and O atoms. |
| Brain/Plasma Ratio (LogBB) | > -1.0 (Kp,br > 0.1) | Direct measure of brain exposure. LogBB = log(Cbrain / Cplasma). | In vivo pharmacokinetic study. |
Advanced Modeling and Interplay of Descriptors
Contemporary models move beyond independent thresholds to multi-parameter equations and machine learning algorithms. A pivotal concept is the Lipophilicity-Polarity Balance.
Key Equation: AlogP98/PSA Model An influential model suggests passive diffusion is optimized when: LogP - (PSA/100) > 0 This heuristic emphasizes the trade-off: lipophilicity must offset polarity.
More sophisticated models like Brain Uptake Index (BUI) or P-gp Substrate Probability integrate these parameters non-linearly. For instance, high MW can be tolerated if accompanied by optimal LogD and low PSA, but the combination of high MW and high PSA is particularly detrimental.
Experimental Protocol: In Vitro BBB Permeability Assay (PAMPA-BBB)
- Objective: High-throughput assessment of passive transmembrane permeability.
- Materials: PAMPA-BBB kit (e.g., pION Inc.), donor plate (lipid-impregnated membrane), acceptor plate, test compound solution, UV plate reader or LC-MS/MS.
- Procedure:
- Prepare donor solution: Compound in pH 7.4 buffer.
- Prepare acceptor solution: Brain-sink buffer (pH 7.4).
- Fill donor plate wells with donor solution.
- Carefully place acceptor plate on top, forming a sandwich.
- Incubate undisturbed for 4-18 hours at 25°C.
- Analyze compound concentration in both donor and acceptor compartments.
- Calculate effective permeability (Pe) using the equation:
Pe = -{ln(1 - [Drug]acceptor / [Drug]equilibrium)} / (A * (1/V_donor + 1/V_acceptor) * t)where A = filter area, t = incubation time, V = volume.
- Interpretation: Compounds with Pe (x 10⁻⁶ cm/s) > 4.0 are considered highly permeable; < 2.0 are poorly permeable.
Diagram 1: Property Interplay in BBB Permeability
Diagram Title: Factors Influencing BBB Permeability Outcome
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Tools for BBB Permeability Research
| Item | Function & Relevance |
|---|---|
| PAMPA-BBB Assay Kit | Pre-formatted plates for high-throughput passive permeability screening. |
| MDCK or MDCK-MDR1 Cell Lines | Canine kidney cells (with/without human MDR1 gene) for modeling transcellular diffusion + active efflux. |
| Primary Brain Endothelial Cells (e.g., hCMEC/D3) | Immortalized human cell line for more physiologically relevant in vitro BBB models. |
| LC-MS/MS System | Gold-standard for quantifying drug concentrations in complex matrices (plasma, brain homogenate). |
| P-gp Substrate Assay (e.g., Calcein-AM) | Fluorescent probe to assess P-glycoprotein efflux activity in cellular models. |
| Molecular Modeling Software (e.g., Schrodinger, MOE) | Computes 3D-PSA, LogP, and other descriptors; runs QSAR/QSPR models. |
| In Vivo Microdialysis Probes | For direct, continuous measurement of free drug concentration in brain interstitial fluid. |
Integrated Workflow for Compound Profiling
A modern approach integrates computational and experimental tiers.
Diagram 2: Integrated BBB Permeability Assessment Workflow
Diagram Title: Tiered Experimental Workflow for BBB Assessment
Successful CNS drug candidates are not defined by a single property but occupy a precise multi-dimensional space. The interplay between MW, lipophilicity, PSA, and HBD/A is dynamic and often compensatory. The future lies in advanced in silico models trained on high-quality in vitro and in vivo data that capture these complex relationships, moving decisively beyond simple rules to enable the rational design of brain-penetrant therapeutics.
From Prediction to Measurement: Tools and Models for Assessing BBB Penetration
The Blood-Brain Barrier (BBB) is a selective, semi-permeable border that protects the central nervous system. For neurotherapeutic drug development, predicting BBB permeability is a critical, early-stage hurdle. Decades of research have established molecular weight (MW) and lipophilicity (commonly measured as logP or logD) as two primary physicochemical determinants of passive diffusion across the BBB. This whitepaper details the in silico models and computational methodologies employed to screen compounds for BBB permeability, specifically within the research context of MW and lipophilicity factors, accelerating the identification of promising CNS drug candidates.
Core Predictive Models and Frameworks
Quantitative Structure-Activity Relationship (QSAR) Models
Traditional QSAR models establish a quantitative correlation between molecular descriptors (e.g., AlogP, topological polar surface area (TPSA), MW, hydrogen bond donors/acceptors) and a biological endpoint, such as logBB (log(Brain/Blood concentration ratio)).
Key Equation (Representative): logBB = a(AlogP) + b(TPSA) + c*(MW) + d Where a, b, c are coefficients derived from regression analysis.
Machine Learning (ML) and Deep Learning (DL) Approaches
Modern frameworks utilize supervised ML algorithms to classify compounds (BBB+ vs BBB-) or regress logBB values using complex, non-linear relationships between a vast array of molecular fingerprints and descriptors.
- Common Algorithms: Random Forest (RF), Support Vector Machine (SVM), Gradient Boosting (e.g., XGBoost), and Deep Neural Networks (DNN).
- Input Features: Extended-connectivity fingerprints (ECFP), Mordred descriptors, VolSurf descriptors, and pre-trained molecular embeddings.
- Typical Workflow: Data Curation → Descriptor Calculation → Feature Selection → Model Training/Validation → Prediction on New Chemical Entities.
Table 1: Historical and Contemporary BBB Permeability Datasets Used for Model Training
| Dataset Name | Approx. Size | Key Endpoint(s) | Primary Descriptors Used | Reference/Year (Representative) |
|---|---|---|---|---|
| Molecules in Drug Bank | ~7,000+ | Binary (BBB+/BBB-) | logP, MW, TPSA, HBD/HBA | (Wishart et al., 2018) |
| B³DB | ~8,000 | logBB, Binary Permeability | ECFP4, RDKit descriptors | (Korolev et al., 2023) |
| Curated BBB Challenge Data | ~2,000 | logPS, logBB | 3D VolSurf, Quantum Chemical | (Mendez et al., 2022) |
Table 2: Performance Metrics of Representative Prediction Models (2020-2024)
| Model Type | Algorithm | Dataset | Accuracy / R² | Key Strengths | Limitations |
|---|---|---|---|---|---|
| Classification | Graph Convolutional Network (GCN) | B³DB | 0.94 (AUC) | Learns spatial structure directly | High computational cost; "black box" |
| Regression (logBB) | XGBoost | Consolidated Set (~5k) | 0.78 (R²) | High interpretability, robust | May miss complex 3D interactions |
| Binary Classifier | Random Forest | DrugBank BBB+/- | 0.89 (Bal Acc) | Handles non-linear relationships | Performance plateaus with size |
Experimental Protocols for Model Development & Validation
Protocol 4.1: Standard QSAR Model Development for logBB Prediction
Objective: To build a multiple linear regression (MLR) model predicting logBB from physicochemical descriptors.
- Data Curation: Obtain a curated dataset of compounds with experimentally measured logBB values (e.g., from published literature). Apply filters for data quality (removing duplicates, outliers).
- Descriptor Calculation: For each SMILES string, calculate 2D molecular descriptors using software like RDKit or PaDEL-Descriptor: Molecular Weight (MW), Octanol-Water Partition Coefficient (AlogP or XlogP), Topological Polar Surface Area (TPSA), Number of Rotatable Bonds (nRotB), Hydrogen Bond Donor/Acceptor count (HBD/HBA).
- Data Splitting: Randomly split the dataset into a training set (70-80%) and a test set (20-30%).
- Model Building: Perform stepwise multiple linear regression on the training set to select the most significant descriptors (e.g., p-value < 0.05). The final model may take the form: Predicted logBB = 0.25(AlogP) - 0.015(TPSA) - 0.002*(MW) + 0.45.
- Validation: Apply the model to the held-out test set. Calculate performance metrics: R² (coefficient of determination), Q² (cross-validated R²), and root mean square error (RMSE).
Protocol 4.2: Machine Learning Pipeline for BBB Permeability Classification
Objective: To train a binary classifier (BBB+ or BBB-) using a Random Forest algorithm.
- Data Preparation: Source a labeled dataset (e.g., from B³DB). Convert SMILES to molecular graphs or fingerprints (ECFP4 with 1024-bit length is standard).
- Feature Engineering: Generate ECFP4 fingerprints for all compounds. Optionally, add key physicochemical descriptors (logP, MW) to the feature vector.
- Dataset Partitioning: Split data into training (70%), validation (15%), and test (15%) sets, ensuring class balance via stratified sampling.
- Model Training: Train a Random Forest classifier (scikit-learn) on the training set. Optimize hyperparameters (nestimators, maxdepth) using the validation set and grid/randomized search with 5-fold cross-validation.
- Evaluation: Predict on the unseen test set. Generate a confusion matrix and report accuracy, precision, recall, F1-score, and Area Under the ROC Curve (AUC-ROC).
Visualizing the Workflow and Relationships
Diagram Title: In Silico BBB Permeability Prediction Workflow
Diagram Title: Core Molecular Factors Impacting BBB Permeability
The Scientist's Toolkit: Essential Research Reagents & Software
Table 3: Key Tools for In Silico BBB Permeability Research
| Tool/Reagent Name | Type | Primary Function in Context | Reference/Source |
|---|---|---|---|
| RDKit | Open-Source Software | Calculates molecular descriptors, fingerprints, and handles cheminformatics operations. | www.rdkit.org |
| PaDEL-Descriptor | Software | Generates >1,800 molecular descriptors and fingerprints from chemical structures. | http://www.yapcwsoft.com/dd/padeldescriptor/ |
| scikit-learn | Python Library | Provides robust implementations of ML algorithms (RF, SVM) for model building. | https://scikit-learn.org |
| DeepChem | Python Library | Offers tools for deep learning on molecular data, including graph neural networks. | https://deepchem.io |
| B³DB Database | Curated Dataset | A benchmark dataset for BBB permeability prediction models (logBB & binary). | https://github.com/theochem/B3DB |
| MoleculeNet/BBBp | Benchmarked Dataset | A curated subset for binary BBB permeability classification tasks. | https://moleculenet.org |
| KNIME Analytics Platform | Workflow Tool | Enables visual assembly of data processing, descriptor calculation, and modeling nodes. | https://www.knime.com |
| Commercial ADMET Predictors | Software Suite | Platforms like Schrödinger's QikProp, Simulations Plus' ADMET Predictor offer proprietary, validated BBB models. | Vendor Specific |
Within the critical research on Blood-Brain Barrier (BBB) permeability factors—focusing on the interplay of molecular weight, lipophilicity, and active transport—the selection and implementation of appropriate in vitro models is paramount. This guide provides a technical deep-dive into three cornerstone methodologies: Transwell assays, the Parallel Artificial Membrane Permeability Assay for the BBB (PAMPA-BBB), and advanced cell-based co-culture systems. Each model offers unique advantages and yields specific data types integral to predicting central nervous system (CNS) drug penetration.
Transwell Assay
The Transwell assay is a workhorse for measuring solute permeability across a confluent monolayer of brain endothelial cells cultured on a semi-permeable membrane insert.
Detailed Protocol
- Cell Seeding: Plate primary human brain microvascular endothelial cells (HBMECs) or immortalized cell lines (e.g., hCMEC/D3) onto the apical side (upper chamber) of a collagen-coated polyester membrane insert (typically 0.4 µm or 1.0 µm pore size, 12-well or 24-well format). A seeding density of 50,000–100,000 cells/cm² is standard.
- Barrier Formation: Culture cells for 3–7 days in endothelial growth medium, often supplemented with physiological hydrocortisone to enhance tight junction formation. Change medium every 48 hours.
- Integrity Validation: Measure Transendothelial Electrical Resistance (TEER) using an epithelial voltohmmeter prior to each experiment. Acceptable monolayers typically show TEER >150 Ω·cm² for hCMEC/D3 and significantly higher for primary cells. Alternatively, validate by measuring the apical-to-basolateral apparent permeability (Papp) of a paracellular integrity marker like sodium fluorescein (376 Da) or FITC-dextran (4 kDa).
- Permeability Experiment: Replace medium in both apical and basolateral compartments with assay buffer (e.g., Hanks' Balanced Salt Solution, HBSS). Add the test compound to the apical donor compartment. Place the plate on an orbital shaker (50-100 rpm) at 37°C to minimize the unstirred water layer.
- Sampling: At predetermined time points (e.g., 30, 60, 90, 120 min), sample aliquots (e.g., 100 µL) from the basolateral acceptor compartment. Replace with fresh pre-warmed buffer to maintain sink conditions.
- Analysis: Quantify compound concentration in samples using HPLC-MS/MS or a plate reader (for fluorescent compounds). Calculate the Papp (cm/s) using the formula: Papp = (dQ/dt) / (A * C₀) where dQ/dt is the steady-state flux rate (mol/s), A is the membrane surface area (cm²), and C₀ is the initial donor concentration (mol/mL).
Table 1: Key Permeability Coefficients from Transwell Models
| Compound (MW, LogP) | Cell Model | Papp (x10⁻⁶ cm/s) | Classification | Reference Standard |
|---|---|---|---|---|
| Caffeine (194, -0.07) | hCMEC/D3 | ~35 | High Permeability | Internal Control |
| Atenolol (266, 0.16) | hCMEC/D3 | ~1.5 | Low Permeability | Paracellular Marker |
| Verapamil (454, 3.8) | hCMEC/D3 | ~25 (Efflux Ratio >2) | P-gp Substrate | Efflux Transporter Control |
| Diazepam (284, 2.99) | hCMEC/D3 | ~50 | High Permeability | Transcellular Passive Diffusion |
| Sucrose (342, -3.7) | Primary Bovine | <1.0 | Very Low Permeability | Integrity/Paracellular Marker |
Diagram Title: Transwell Assay Experimental Workflow
PAMPA-BBB
PAMPA-BBB is a non-cell-based, high-throughput screen that predicts passive transcellular diffusion through a lipid-infused artificial membrane.
Detailed Protocol
- Plate Preparation: Use a 96-well PAMPA sandwich plate. The donor plate is a standard well plate. The acceptor plate has a microfilter on its bottom.
- Membrane Formation: Add 5 µL of a porcine brain lipid extract (PBL) in dodecane (e.g., 20 mg/mL) to each filter of the acceptor plate. The lipid forms a thin, artificial bilayer.
- Assay Buffer: Fill the donor (apical) wells with 150-300 µL of a pH 7.4 phosphate buffer containing the test compound (typically 50-100 µM). Fill the acceptor (basolateral) wells with 300 µL of pH 7.4 buffer.
- Assembling the Sandwich: Carefully place the acceptor plate on top of the donor plate, ensuring the lipid-coated filter is in contact with the donor solution. Avoid air bubbles.
- Incubation: Incubate the assembled plate at 25°C (room temperature) for 4-18 hours without agitation.
- Analysis: Disassemble the plate. Quantify the compound concentration in both donor and acceptor wells using UV spectroscopy (direct concentration measurement if no UV chromophore) or LC-MS/MS.
- Calculation: Determine the effective permeability, Pe (x10⁻⁶ cm/s), using a validated equation accounting for membrane retention.
Table 2: PAMPA-BBB Permeability Benchmarks
| Compound | Pe (x10⁻⁶ cm/s) PAMPA-BBB | BBB Permeability Prediction | Category (MW, LogP) |
|---|---|---|---|
| Testosterone (288, 3.32) | ~17.0 | CNS+ (High) | High Lipophilicity |
| Propranolol (259, 3.48) | ~8.5 | CNS+ | Moderate Lipophilicity |
| Corticosterone (346, 1.94) | ~5.5 | CNS+/- (Borderline) | Moderate Lipophilicity |
| Hydrocortisone (362, 1.61) | ~1.2 | CNS- (Low) | Low Lipophilicity |
| Prazosin (383, 2.50)* | ~0.8 | CNS- (Underpredicts) | P-gp Substrate |
*PAMPA underpredicts permeability for strong efflux substrates as it lacks transporters.
Diagram Title: PAMPA-BBB Sandwich Plate Setup
Cell-Based Co-culture Systems
These advanced models incorporate astrocytes, pericytes, or neurons in co-culture with brain endothelial cells to induce a more physiologically relevant BBB phenotype via cell-cell signaling.
Detailed Protocol (Astrocyte-Endothelial Co-culture)
- Astrocyte Culture: Plate primary rat or human astrocytes onto the bottom of a multi-well plate (or on the basolateral side of the membrane for contact models). Culture until confluent.
- Endothelial Cell Seeding: For a non-contact (paracrine) model, plate endothelial cells onto collagen/fibronectin-coated Transwell inserts as in Section 1.
- Co-culture Assembly: Place the endothelial cell-seeded insert into the well containing the confluent astrocyte layer. The cells share medium but are not in physical contact.
- Culture Conditions: Maintain in specialized co-culture medium (e.g., endothelial basal medium with astrocyte-conditioned medium, or a 1:1 mix of respective media) for 5-7 days to allow inductive signaling.
- Characterization: Validate with elevated TEER (>300-500 Ω·cm² for hCMEC/D3), reduced marker permeability, and upregulated expression of tight junction proteins (claudin-5, occludin) and efflux transporters (P-gp, BCRP) via qPCR or immunostaining.
- Functional Assays: Perform permeability assays as in Section 1. Include specific inhibitors (e.g., Cyclosporin A for P-gp) to assess active efflux contribution.
Table 3: Impact of Co-culture on BBB Properties
| Measured Parameter | Mono-culture (Endothelial Only) | Co-culture (Endothelial + Astrocytes) | Functional Implication |
|---|---|---|---|
| Typical TEER (Ω·cm²) | 50 - 200 | 200 - 800+ | Enhanced Barrier Tightness |
| Sucrose Papp (x10⁻⁶ cm/s) | ~2.0 - 4.0 | ~0.5 - 1.5 | Reduced Paracellular Leak |
| P-gp Activity (Efflux Ratio) | Moderate (2-5) | High (5-20) | Induced Active Transport |
| Alkaline Phosphatase Activity | Low | High (~3-5 fold increase) | Induction of BBB Enzymes |
Diagram Title: Co-culture Signaling & BBB Induction
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function & Rationale |
|---|---|
| Primary HBMECs or Immortalized Lines (hCMEC/D3, RBE4) | Provide a biologically relevant endothelial base expressing key BBB transporters and junctional proteins. Choice depends on throughput vs. physiological fidelity. |
| Porcine Brain Lipid Extract (PBL) | Key component of PAMPA-BBB membranes, mimicking the lipid composition of the endothelial plasma membrane to predict passive diffusion. |
| Collagen Type IV & Fibronectin | Essential extracellular matrix proteins for coating Transwell membranes, promoting endothelial cell adhesion, spreading, and barrier formation. |
| Hydrocortisone / Dexamethasone | Glucocorticoids used in culture media to enhance tight junction assembly and increase TEER in both mono- and co-culture models. |
| Sodium Fluorescein or FITC-Dextran (4 kDa) | Integrity markers. Their low passive permeability allows validation of monolayer confluence and tight junction competence before permeability assays. |
| Reference Compounds Kit (Caffeine, Atenolol, Verapamil, Rhodamine 123) | A standardized set of high, low, and efflux-substrate permeability controls essential for model calibration and inter-experiment comparison. |
| TEER Measurement System (e.g., Epithelial Voltohmmeter) | Critical for non-destructive, quantitative assessment of barrier integrity and maturation over time. |
| P-gp/BCRP Specific Inhibitors (e.g., Elacridar, Ko143) | Used in permeability assays to pharmacologically block specific efflux transporters, enabling calculation of their contribution to net flux. |
| Astrocyte-Conditioned Medium | Contains astrocyte-derived trophic factors; used in non-contact co-culture models to induce and maintain a robust BBB phenotype in endothelial cells. |
The assessment of a compound's ability to cross the blood-brain barrier (BBB) is a critical step in central nervous system (CNS) drug development. The permeability of the BBB is governed by complex physical and biochemical factors, with molecular weight (MW) and lipophilicity (often expressed as Log P or Log D) being primary determinants. This technical guide details the core in vitro and in vivo experimental metrics used to quantify BBB penetration: Apparent Permeability (Papp or Pe), Efflux Ratio (ER), and the unbound brain-to-plasma concentration ratio (Kp,uu). These metrics are indispensable for understanding a molecule's passive diffusion potential, its susceptibility to active efflux transporters (e.g., P-glycoprotein/P-gp, BCRP), and its ultimate free concentration at the target site.
Core Experimental Metrics: Definitions and Significance
Apparent Permeability (Pe)
Definition: A measure of the rate of compound translocation across a cellular monolayer in an in vitro BBB model, typically reported in units of cm/s × 10-6. It reflects the combined effects of passive transcellular diffusion and paracellular leakage. Significance: High Pe (>10-15 × 10-6 cm/s) generally indicates good passive permeability, a prerequisite for CNS penetration.
Efflux Ratio (ER)
Definition: Calculated as Pe(B-to-A) / Pe(A-to-B) in a directional permeability assay. An ER > 2-3 suggests active efflux transport. Significance: Identifies substrates of efflux transporters (e.g., P-gp). A high ER is a major liability for CNS drugs, as it actively restricts brain entry.
Unbound Brain-to-Plasma Concentration Ratio (Kp,uu)
Definition: The gold-standard in vivo metric for brain penetration. Kp,uu = (Cbrain, unbound / Cplasma, unbound). It represents the net result of all processes at the BBB: passive diffusion, active influx, and active efflux. Significance: A Kp,uu ~1 indicates equilibrium of unbound drug between plasma and brain. Kp,uu < 1 suggests net efflux, while Kp,uu > 1 suggests net active uptake.
Table 1: Interpretation of Key BBB Permeability Metrics
| Metric | Typical Range | Interpretation for CNS Drug Development |
|---|---|---|
| Pe (× 10-6 cm/s) | < 1 | Low permeability (poor passive diffusion) |
| 1 - 10 | Moderate permeability | |
| > 10 - 15 | High permeability (favorable for passive diffusion) | |
| Efflux Ratio (ER) | < 2 | Not an efflux transporter substrate |
| 2 - 10 | Moderate efflux substrate | |
| > 10 | Strong efflux substrate (significant liability) | |
| Kp,uu | << 1 (e.g., 0.1) | Net efflux, low free brain exposure |
| ~ 0.3 - 3 | Moderate to good free brain exposure | |
| ~ 1 | Equilibrium achieved (ideal for most targets) | |
| >> 1 | Net active uptake into brain |
Table 2: Impact of Molecular Properties on BBB Metrics (General Trends)
| Molecular Property | Impact on Pe | Impact on ER (P-gp risk) | Impact on Kp,uu |
|---|---|---|---|
| High Lipophilicity (Log D > 3) | Increases (but can plateau/decline) | Often increases | May decrease due to binding or efflux |
| Optimal Log D (~2-3) | Maximizes passive diffusion | Often minimized | Tends to optimize towards ~1 |
| High MW (>450 Da) | Decreases (paracellular restricted) | Can increase | Often decreases |
| H-Bond Donors >3 | Decreases | Can increase | Often decreases |
Detailed Experimental Protocols
1In VitroMDCK or hCMEC/D3 Assay for Peand ER
Purpose: To determine apparent permeability and identify efflux transporter substrates. Protocol:
- Cell Culture: Seed Madin-Darby Canine Kidney (MDCKII) cells overexpressing human MDR1 (P-gp) or human cerebral microvascular endothelial cells (hCMEC/D3) onto collagen-coated transwell inserts. Culture for 7-10 days to form confluent, tight monolayers (TEER > 150 Ω·cm²).
- TEER Measurement: Measure Trans-Endothelial Electrical Resistance (TEER) prior to experiment to confirm monolayer integrity.
- Compound Preparation: Prepare test compound (typical 2-10 µM) in transport buffer (e.g., HBSS with 10 mM HEPES, pH 7.4). Include a high-permeability control (e.g., propranolol) and a low-permeability control (e.g., atenolol).
- Bidirectional Transport:
- A-to-B (Apical-to-Basolateral): Add compound to donor (apical) chamber. Sample from receiver (basolateral) chamber at e.g., 30, 60, 90, and 120 minutes.
- B-to-A (Basolateral-to-Apical): Add compound to donor (basolateral) chamber. Sample from receiver (apical) chamber at the same time points.
- Inhibition Assay (Optional): Repeat transport in the presence of a potent efflux inhibitor (e.g., 1-5 µM zosuquidar for P-gp) to confirm transporter involvement.
- LC-MS/MS Analysis: Quantify compound concentration in all samples.
- Calculations:
- Papp (cm/s) = (dQ/dt) / (A * C0), where dQ/dt is the flux rate, A is the membrane area, and C0 is the initial donor concentration.
- Efflux Ratio = Papp (B-to-A) / Papp (A-to-B)
2In VivoRodent Brain Exposure Study for Kp,uu
Purpose: To determine the unbound brain-to-plasma concentration ratio in vivo. Protocol:
- Dosing and Sampling: Administer test compound to rodents (rat/mouse) via intended route (e.g., IV bolus, PO). At predetermined time points (e.g., 0.5, 2, 6h), collect terminal blood (into heparinized tubes) and whole brain.
- Plasma Processing: Centrifuge blood to obtain plasma.
- Brain Homogenate Preparation: Weigh brain tissue, homogenize in buffer (e.g., 3-4 volumes of phosphate buffer). An aliquot is taken for total brain concentration (Cbrain,total) analysis.
- Determination of Unbound Fractions (fu):
- Plasma fu (fu,plasma): Use rapid equilibrium dialysis (RED) or ultracentrifugation. Spike compound into blank plasma, place in donor chamber separated by a dialysis membrane from buffer. After equilibrium (~4-6h, 37°C), quantify concentrations on both sides. fu,plasma = [Buffer]/[Plasma].
- Brain fu (fu,brain): Use brain homogenate (not tissue) in a similar RED setup. fu,brain = [Buffer]/[Homogenate]. Note: This value must be corrected for dilution: fu,brain,corrected = 1 / (D + (1-D)/fu,brain,measured), where D is the dilution factor.
- Bioanalysis: Use LC-MS/MS to determine total concentrations in plasma, brain homogenate, and all dialysis samples.
- Calculations:
- Cbrain,unbound = fu,brain,corrected × Cbrain,total
- Cplasma,unbound = fu,plasma × Cplasma,total
- Kp,uu = Cbrain,unbound / Cplasma,unbound
- (Note: Total Kp = Cbrain,total / Cplasma,total is a misleading metric and should not be used alone).
Visualizations and Workflows
Diagram Title: Integrated Workflow for Key BBB Permeability Metrics
Diagram Title: Factors Governing Kp,uu at the Blood-Brain Barrier
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for BBB Permeability Studies
| Item | Function & Application | Example Product/Cell Line |
|---|---|---|
| MDCKII-MDR1 Cells | Standard in vitro model for assessing permeability & P-gp mediated efflux. | NIH MDCKII-MDR1 (NCI), commercially available from vendors. |
| hCMEC/D3 Cells | Immortalized human brain endothelial cell line; more physiologically relevant model. | Merck Millipore, Sigma-Aldrich. |
| Transwell Plates | Permeable supports (e.g., 0.4 µm pore polyester) for growing cell monolayers. | Corning, Greiner Bio-One. |
| EVOM Voltohmmeter | For measuring Trans-Endothelial Electrical Resistance (TEER) to confirm monolayer integrity. | World Precision Instruments. |
| P-gp Inhibitor (Potent) | To confirm efflux transporter involvement in inhibition assays. | Zosuquidar (LY335979), Elacridar (GF120918). |
| Rapid Equilibrium Dialysis (RED) Device | High-throughput method for determining unbound fraction (fu) in plasma and brain homogenate. | Thermo Fisher Scientific RED Plate. |
| LC-MS/MS System | Essential for sensitive and specific quantification of drugs in biological matrices (plasma, brain, buffer). | Various (Sciex, Agilent, Waters, Thermo). |
| Animal Strain (Rodent) | For in vivo pharmacokinetic and brain exposure studies. | Sprague-Dawley rats, CD-1 mice. |
The Critical Role of Plasma Protein Binding and Brain Tissue Binding
Within the broader thesis on Blood-Brain Barrier (BBB) permeability factors—notably molecular weight and lipophilicity—plasma protein binding (PPB) and brain tissue binding (BTB) emerge as critical, often confounding, determinants of central nervous system (CNS) drug disposition. While Log P/D and molecular weight provide initial permeability estimates, they fail to predict unbound drug concentration, the sole driver of pharmacodynamic activity. This whitepaper provides an in-depth technical analysis of PPB and BTB, detailing their experimental determination, impact on pharmacokinetic/pharmacodynamic (PK/PD) relationships, and integration into modern CNS drug discovery paradigms.
Quantitative Foundations: Key Parameters and Impact
The free drug hypothesis posits that only the unbound fraction (fu) of a drug in plasma (fu,p) and brain (fu,brain) is capable of engaging pharmacological targets or crossing membranes. The critical parameter linking systemic exposure to brain exposure is the unbound brain-to-plasma concentration ratio (Kp,uu), defined as:
Kp,uu = (Cu,brain) / (Cu,plasma) = (Ctotal,brain × fu,brain) / (Ctotal,plasma × fu,plasma)
Where:
- Cu,brain = Unbound brain concentration
- Cu,plasma = Unbound plasma concentration
- fu,brain = Fraction unbound in brain tissue
- fu,plasma = Fraction unbound in plasma
Table 1: Impact of PPB and BTB on Key Neuropharmacokinetic Parameters
| Parameter | Definition | Influence of High PPB (Low fu,p) | Influence of High BTB (Low fu,brain) | Ideal Target for CNS Drugs* |
|---|---|---|---|---|
| Vd (Volume of Distribution) | Theoretical volume to contain total drug at plasma concentration. | Increases (drug sequestered in plasma). | Markedly increases (drug distributed into tissue). | Large (indicative of tissue penetration). |
| Clearance (CL) | Volume of plasma cleared of drug per unit time. | Decreases for restrictively cleared drugs (only unbound fraction is cleared). | Minimal direct effect. | Moderate to low. |
| Total Brain/Plasma Ratio (Kp) | Ctotal,brain / Ctotal,plasma. | Can appear artificially high. | Can appear artificially low. | Not indicative alone. |
| Unbound Brain/Plasma Ratio (Kp,uu) | Cu,brain / Cu,plasma. | No direct effect (ratio of unbound concentrations). | No direct effect (ratio of unbound concentrations). | ~1.0 (passive diffusion equilibrium). <1.0 indicates active efflux (e.g., P-gp). >1.0 indicates active uptake. |
| Receptor Occupancy (RO) | Driven by Cu,brain. | Reduces Cu,plasma for a given total dose, potentially lowering Cu,brain if Kp,uu is fixed. | Reduces Cu,brain for a given total brain concentration, requiring higher dosing. | Directly proportional to Cu,brain. |
*Target values assume passive diffusion is the primary mechanism.
Experimental Protocols
Determination of Plasma Protein Binding (fu,p)
Method: Equilibrium Dialysis (Gold Standard) Principle: Separation of protein-bound and unbound drug fractions at equilibrium across a semi-permeable membrane.
Detailed Protocol:
- Preparation: Hydrate a semi-permeable membrane (e.g., cellulose, 12-14 kDa MWCO) in dialysis buffer (e.g., 67 mM phosphate buffer, pH 7.4). Load the Teflon dialysis cell.
- Sample Loading: To one chamber (donor), add plasma (or serum) spiked with the test compound (typically 1-5 µM). To the opposing chamber (receiver), add an equal volume of dialysis buffer.
- Equilibration: Seal cells and rotate in a temperature-controlled incubator at 37°C for 4-6 hours (time must be validated for compound stability).
- Sampling & Analysis: Post-incubation, aliquot samples from both chambers. Analyze drug concentrations using LC-MS/MS.
- Calculation:
- fu,p = (Creceiver) / (Cdonor_initial) Note: Due to potential volume shifts, correction methods (e.g., using a non-permeating marker) are recommended.
- % Bound = (1 - fu,p) × 100.
Alternative Methods: Ultrafiltration, Ultracentrifugation.
Determination of Brain Tissue Binding (fu,brain)
Method: Brain Homogenate Equilibrium Dialysis Principle: Similar to PPB, but using diluted brain homogenate to mimic the intracellular and membrane binding environment.
Detailed Protocol:
- Homogenate Preparation: Fresh or freshly frozen brain tissue is homogenized in buffer (e.g., pH 7.4 phosphate) to a standardized dilution (typically 1:3 or 1:4 brain:buffer, w/v) using a rotor-stator homogenizer.
- Dialysis: Follow the equilibrium dialysis protocol above, substituting plasma with the brain homogenate.
- Calculation & Correction: fu,brain is calculated from the measured fu in homogenate (fu,hom) and corrected for dilution:
- fu,brain = 1 / ( D * (1/fu,hom - 1) + 1 )
- Where D is the dilution factor (e.g., 4 for a 1:4 dilution).
Signaling Pathways and Disposition Relationships
Diagram 1: PK/PD Pathway Integrating PPB & BTB
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagent Solutions for PPB/BTB Studies
| Item | Function & Specification | Example Vendor/Product |
|---|---|---|
| Equilibrium Dialysis Device | Multi-cell apparatus for high-throughput fu determination. | HTDialysis HTD96b, Thermo Fisher Rapid Equilibrium Dialysis (RED) plates. |
| Dialysis Membrane | Semi-permeable barrier separating bound/unbound fractions. Standard MWCO 12-14 kDa. | Spectra/Por 2 or similar regenerated cellulose membranes. |
| Blank Plasma/Serum | Matrix for PPB assays. Species-specific (human, rat, mouse, etc.). | BioreclamationIVT, Sigma-Aldrich, or in-house collection with ethical approval. |
| Dialysis Buffer (pH 7.4) | Isotonic buffer to maintain physiological pH and osmolarity in receiver chamber. | 0.1M phosphate buffer, 0.15M KCl, or Dulbecco's PBS. |
| Brain Homogenization Buffer | Buffer for preparing consistent brain homogenate for BTB assays. | 0.1M phosphate buffer (pH 7.4) or isotonic sucrose solution. |
| Stable Isotope-Labeled Internal Standards | For accurate LC-MS/MS quantification, correcting for matrix effects and recovery. | Compound-specific, often synthesized in-house or by vendors like Alsachim, TLC PharmaChem. |
| Protein-Binding Control Compounds | Validates assay performance. High (e.g., warfarin, ~99% bound) and low (e.g., caffeine, ~30% bound) binding controls. | Commercially available analytical standards. |
| LC-MS/MS System | Gold standard for sensitive and specific quantification of drugs in complex matrices (plasma, buffer, homogenate). | Systems from Sciex, Waters, Agilent, or Thermo Fisher. |
Diagram 2: Integrated PPB/BTB Experimental Workflow
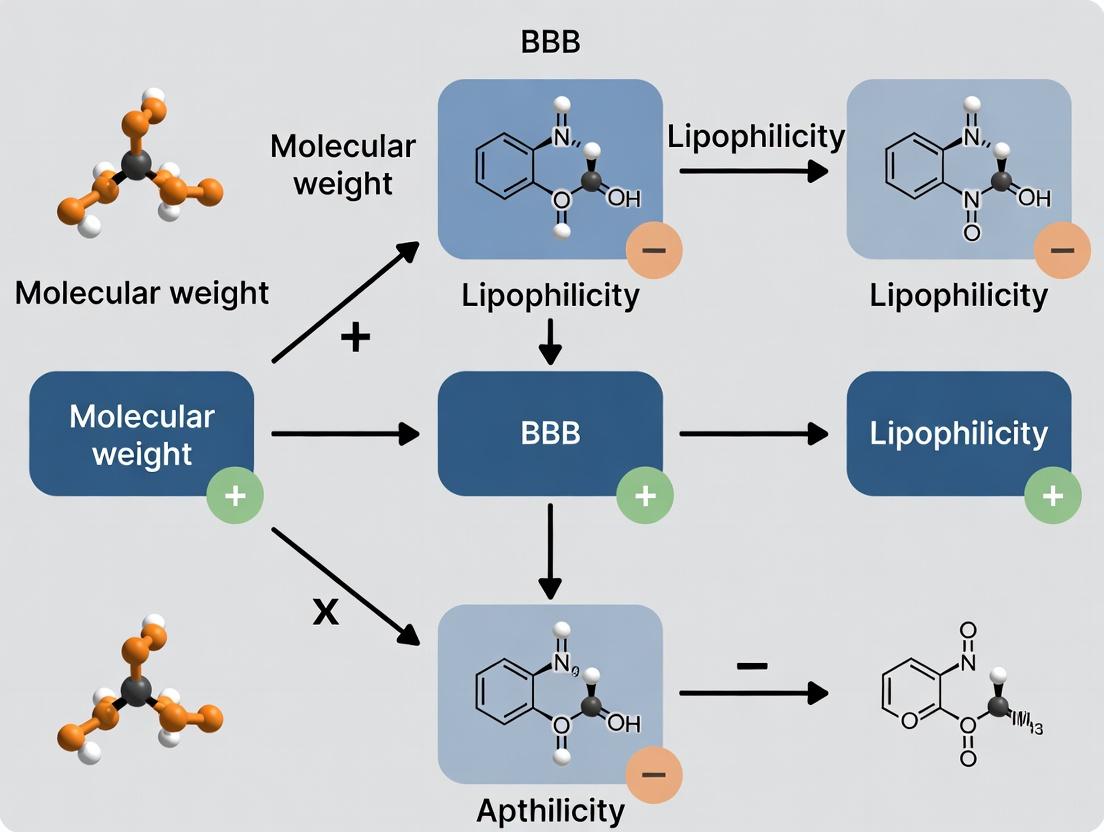
The blood-brain barrier (BBB) presents a formidable challenge in central nervous system (CNS) drug development. Its selective permeability is governed by a complex interplay of physicochemical properties, with molecular weight (MW) and lipophilicity (commonly expressed as LogP) serving as two paramount predictors. This guide details the systematic integration of these key parameters into medicinal chemistry cycles to enhance the probability of achieving sufficient CNS exposure.
Current research, supported by analyses of approved CNS drugs, establishes clear, though not absolute, boundaries. Optimal BBB permeability is typically associated with MW < 450 Da and a calculated LogP (cLogP) or measured LogD (at physiological pH 7.4) in the range of 1–3. Exceeding these limits often leads to poor passive diffusion, increased efflux by P-glycoprotein (P-gP), and/or elevated metabolic clearance.
Core Physicochemical Property Guidelines and Data
The following table summarizes the quantitative targets and associated risks for MW and LogP in CNS-oriented design.
Table 1: MW and LogP Targets for BBB Permeability
| Property | Optimal Range (Target) | Acceptable Range | High-Risk Zone | Primary Consequence of Exceeding Limit |
|---|---|---|---|---|
| Molecular Weight | < 450 Da | 450 - 500 Da | > 500 Da | Drastically reduced passive diffusion; increased P-gP efflux likelihood. |
| LogP (cLogP) | 2.0 - 3.0 | 1.0 - 3.5 | < 1.0 or > 3.5 | Poor membrane permeability (low LogP); poor aqueous solubility, metabolic instability, toxicity (high LogP). |
| LogD₇.₄ | 1.0 - 3.0 | 0.5 - 3.5 | < 0.5 or > 4.0 | More accurate than LogP for ionizable compounds; directly correlates with permeability. |
| TPSA | < 60-70 Ų | 70 - 90 Ų | > 90 Ų | Correlates with hydrogen bonding capacity; impacts passive diffusion. |
Experimental Protocols for Key Measurements
Protocol for Determining LogP/LogD via Shake-Flask Method
Principle: The distribution of a compound between immiscible aqueous and organic phases (typically n-octanol and buffer) at equilibrium.
Procedure:
- Solution Preparation: Pre-saturate n-octanol with phosphate buffer (pH 7.4) and vice-versa overnight. Dissolve the test compound in the pre-saturated phase where it is more soluble (typically ~0.5-1 mg/mL).
- Partitioning: Combine equal volumes (e.g., 1 mL each) of the compound solution in one phase and the pre-saturated counterpart phase in a glass vial. Cap tightly.
- Equilibration: Shake vigorously for 1 hour at constant temperature (e.g., 25°C). Allow phases to separate completely for several hours or centrifuge.
- Quantification: Carefully separate the two phases. Analyze the concentration of the compound in each phase using a validated analytical method (e.g., HPLC-UV, LC-MS/MS).
- Calculation: LogD₇.₄ = Log₁₀ (Concentration in n-octanol / Concentration in buffer). For non-ionizable compounds, this equals LogP.
Protocol for In Vitro BBB Permeability Assessment (PAMPA-BBB)
Principle: Parallel Artificial Membrane Permeability Assay (PAMPA) models passive diffusion across the BBB using a specialized lipid membrane.
Procedure:
- Membrane Preparation: Coat a hydrophobic filter on a 96-well plate with a 2% (w/v) solution of porcine brain lipid in dodecane (e.g., 5 µL/well). Allow solvent to evaporate briefly to form the artificial membrane.
- Plate Assembly: Fill the acceptor plate (bottom) with PBS buffer (pH 7.4). Place the donor plate (containing the lipid membrane) on top.
- Dosing: Add a solution of test compound (50-100 µM) in PBS pH 7.4 to the donor wells.
- Incubation: Assemble the sandwich plate and incubate undisturbed at room temperature for 4-18 hours.
- Analysis: Quantify compound concentration in both donor and acceptor wells via LC-MS/MS.
- Calculation: Determine the effective permeability, Pₑ (×10⁻⁶ cm/s). Compounds with Pₑ > 3.0-4.0 ×10⁻⁶ cm/s are considered highly permeable.
Visualization of the Design and Decision Cycle
Diagram 1: MW/LogP Integration Cycle in Medicinal Chemistry
The Scientist's Toolkit: Key Research Reagents and Materials
Table 2: Essential Reagents for MW/LogP and Permeability Studies
| Item | Function / Application | Example / Specification |
|---|---|---|
| n-Octanol | Organic solvent for shake-flask LogP/LogD determination; mimics the lipid environment. | HPLC grade, pre-saturated with appropriate buffer. |
| Porcine Brain Lipid Extract | Critical component for constructing the artificial membrane in PAMPA-BBB assays. | Commercial PAMPA-BBB kits (e.g., pION Inc., BD Gentest). |
| PAMPA Plate Assemblies | Multi-well plates designed for permeability screening with donor and acceptor compartments. | 96-well format, polycarbonate or PVDF filter plates. |
| pH 7.4 Phosphate Buffer | Aqueous phase for LogD₇.₄ and PAMPA assays, simulating physiological pH. | 0.1 M phosphate buffer, ionic strength adjusted. |
| LC-MS/MS System | Gold-standard for sensitive and specific quantification of compound concentrations in complex matrices. | Systems from Agilent, Waters, Sciex, or Thermo Fisher. |
| Molecular Modeling Software | Calculate cLogP, MW, TPSA, and other descriptors; visualize SAR. | Schrödinger Suite, MOE, ChemAxon, OpenEye toolkits. |
| Reference Compounds | Validation of assay performance with known high, medium, and low permeability compounds. | Caffeine, Verapamil (high); Ranitidine (low). |
Overcoming the Barrier: Strategies for Optimizing Brain Exposure of Challenging Molecules
In the pursuit of central nervous system (CNS) therapeutics, crossing the blood-brain barrier (BBB) is a paramount challenge. This whitepaper examines the critical role of lipophilicity within a broader thesis on BBB permeability factors, which also encompass molecular weight, hydrogen bonding, and charge. Lipophilicity, most often quantified as Log P (octanol-water partition coefficient) or Log D (distribution coefficient at physiological pH), represents a fundamental molecular property that must be optimized to navigate a delicate balance: sufficient lipid character to passively diffuse across biological membranes, yet adequate aqueous solubility for dissolution and absorption, while avoiding excessive metabolic clearance. Identifying this 'sweet spot' is a cornerstone of modern CNS drug design.
Quantitative Landscape of Lipophilicity in Drug Properties
Lipophilicity is a key driver of multiple ADME (Absorption, Distribution, Metabolism, Excretion) properties. The following tables summarize critical quantitative relationships and guidelines.
Table 1: Impact of Log P / Log D on Key Drug Properties
| Property | Optimal Range (General) | Optimal Range (CNS Focus) | Consequence of High Log P | Consequence of Low Log P |
|---|---|---|---|---|
| Permeability | Log P ~1-4 | Log D~(1.5-3.0)* | Increased passive diffusion | Poor membrane permeation |
| Aqueous Solubility | Log P <3 | Log D <3 | Poor solubility, formulation challenges | Good solubility |
| Metabolic Clearance | Log P <3 | Log D <3 | Increased CYP450 metabolism, poor stability | Reduced metabolic clearance |
| Protein Binding | Log P <4 | Log D <3 | High non-specific binding, reduced free fraction | Low binding, high free fraction |
| hERG Inhibition Risk | Log P <4 | Log D <3 | Increased risk of cardiac toxicity | Reduced hERG risk |
Note: CNS drugs often target the higher end of this range for sufficient BBB penetration. (Sources: Wager et al., *ACS Chem. Neurosci., 2016; Rankovic, J. Med. Chem., 2015)*
Table 2: Lipophilicity Guidelines from Key Studies
| Study / Metric | Recommended Range | Key Finding / Rationale |
|---|---|---|
| Lipinski's Rule of 5 | Log P ≤5 | Oral druglikeness filter. |
| "Golden Triangle" | Log P 1-3, MW 200-400 | Optimal balance of developability properties (Johnson et al.). |
| CNS MPO Score | Log P/Log D contributes to a multi-parameter score (Target >4) | A desirability score where Log D (pH 7.4) ideally 1-3 (Wager et al.). |
| Fraction Unbound in Brain (fu,brain) | Favored by moderate Log D | High Log D leads to high nonspecific brain tissue binding, reducing available free drug. |
Experimental Protocols for Key Determinations
Determination of Log P / Log D (Shake-Flask Method)
Principle: The distribution of a compound between octanol (organic phase) and aqueous buffer (e.g., phosphate buffer, pH 7.4) is measured at equilibrium. Protocol:
- Preparation: Saturate 1-octanol and buffer phase with each other overnight. Prepare a stock solution of the test compound in either phase or DMSO (≤1% final).
- Partitioning: Combine equal volumes (e.g., 0.5 mL each) of octanol and buffer in a glass vial. Add compound. Cap tightly.
- Equilibration: Shake vigorously for 1 hour at constant temperature (e.g., 25°C). Centrifuge to separate phases clearly.
- Quantification: Carefully sample each layer. Analyze compound concentration in each phase using a validated method (e.g., HPLC-UV, LC-MS).
- Calculation: Log D = log10([Compound]octanol / [Compound]buffer). For Log P, use pH where compound is non-ionized.
Parallel Artificial Membrane Permeability Assay (PAMPA)
Principle: A high-throughput assay modeling passive transcellular permeability using an artificial lipid membrane. Protocol:
- Membrane Formation: Prepare a lipid solution (e.g., 2% w/v phosphatidylcholine in dodecane). Add to a filter on a 96-well acceptor plate.
- Plate Setup: Place the acceptor plate on top of a donor plate containing buffer (e.g., PBS, pH 7.4). Fill acceptor wells with buffer.
- Dosing: Add test compound to donor wells. Include high-permeability (e.g., propranolol) and low-permeability (e.g., atenolol) controls.
- Incubation: Seal the plate sandwich and incubate for 2-6 hours undisturbed.
- Analysis: Quantify compound in both donor and acceptor compartments by UV plate reader or LC-MS.
- Calculation: Determine effective permeability (Pe) using a standard equation accounting for sink conditions.
Metabolic Stability Assay (Microsomal Incubation)
Principle: Measures the intrinsic clearance of a compound by liver microsomal enzymes (e.g., CYP450s). Protocol:
- Incubation Mix: Prepare reaction mix containing human or rat liver microsomes (0.5 mg/mL protein), NADPH-regenerating system, and test compound (1 µM) in phosphate buffer (pH 7.4).
- Initiation: Start reaction by adding NADPH system or microsomes. Run in triplicate.
- Time Course: Aliquot samples at time points (e.g., 0, 5, 15, 30, 45, 60 min) into acetonitrile to stop reaction.
- Sample Processing: Centrifuge to precipitate proteins. Analyze supernatant by LC-MS/MS.
- Data Analysis: Plot remaining compound (%) vs. time. Calculate half-life (t1/2) and intrinsic clearance (CLint).
Visualization of Concepts and Workflows
Diagram 1: The Lipophilicity Balancing Act
Diagram 2: Lipophilicity Optimization Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Lipophilicity & Permeability Studies
| Reagent / Material | Supplier Examples | Function in Research |
|---|---|---|
| 1-Octanol (HPLC Grade) | Sigma-Aldrich, Millipore | Organic phase for definitive Log P/D shake-flask experiments. |
| Pre-coated PAMPA Plates (e.g., PAMPA-BBB) | pION, Corning | Ready-to-use plates for high-throughput permeability screening with BBB-specific lipid formulations. |
| Human/Rat Liver Microsomes | Corning, Xenotech | Source of metabolic enzymes (CYP450) for intrinsic clearance studies. |
| NADPH Regenerating System | Promega, Corning | Provides essential cofactor for cytochrome P450 activity in stability assays. |
| Phosphatidylcholine (e.g., Egg Lecithin) | Avanti Polar Lipids, Sigma | Key lipid for creating artificial membranes in custom PAMPA or vesicle studies. |
| Biorelevant Dissolution Media (FaSSIF/FeSSIF) | Biorelevant.com, USP | Simulates intestinal fluids for more predictive solubility and permeability measurements. |
| Reference Compounds (Propranolol, Atenolol, Warfarin) | Sigma-Aldrich, Tocris | High and low permeability/specific binding controls for assay validation. |
| LC-MS/MS Systems & Columns | Agilent, Waters, Shimadzu | Essential for sensitive and specific quantification of compounds in complex matrices from ADME assays. |
MW Creep and Strategies for Molecular Weight Reduction in Lead Optimization
Within the critical path of drug discovery, achieving sufficient Blood-Brain Barrier (BBB) permeability is a formidable challenge, governed by a delicate interplay of key physicochemical properties. The broader research thesis on BBB permeability factors—molecular weight (MW), lipophilicity (often expressed as LogP or LogD), hydrogen bonding potential, and polar surface area—identifies MW as a primary driver of passive diffusion. "Molecular Weight Creep" (MW Creep) refers to the systematic and often inadvertent increase in a lead compound's molecular weight during optimization, typically as functional groups are added to improve potency or selectivity. This escalation directly antagonizes the goal of enhancing CNS penetration, as BBB permeability inversely correlates with molecular size. This guide details the causes of MW creep and presents actionable, evidence-based strategies for molecular weight reduction, framed within the imperative of optimizing for the BBB permeability landscape.
The Problem of MW Creep: Causes and Consequences
MW creep typically originates from iterative medicinal chemistry cycles where adding bulky substituents is the most straightforward path to address specific shortcomings.
Primary Drivers:
- Potency Optimization: Adding hydrophobic or aromatic moieties to fill binding pockets.
- Metabolic Stability: Introducing blocking groups (e.g., deuterium, fluoro, methyl) on metabolically soft spots.
- Solubility Enhancement: Appending ionizable groups or polyethylene glycol (PEG) chains.
- Selectivity Improvement: Adding specific functional groups to disfavor off-target binding.
Quantitative Impact on BBB Penetration: Data from in vivo and in vitro BBB models consistently show a sharp decline in the probability of brain penetration as MW increases beyond optimal ranges.
Table 1: Correlation of Molecular Weight with Key BBB Permeability Metrics
| Molecular Weight Range (Da) | P-gp Efflux Ratio (Typical) | Log PS (Permeability-Surface Area Product)* | % Probability of High CNS Penetration (Historical Dataset) |
|---|---|---|---|
| < 300 | Low (< 2.0) | > -2.5 | > 85% |
| 300 - 400 | Moderate (2.0 - 5.0) | -2.5 to -3.5 | 50% - 85% |
| 400 - 500 | High (> 5.0) | -3.5 to -4.5 | 10% - 50% |
| > 500 | Very High (> 10.0) | < -4.5 | < 10% |
Log PS data adapted from preclinical *in situ perfusion studies. Permeability decreases by approximately one log order per ~150-200 Da increase.
Strategic Framework for Molecular Weight Reduction
The following strategies should be employed iteratively and in parallel with potency and ADMET assays.
Scrutinize and Prune the Scaffold (Core Replacement)
Evaluate if the core scaffold itself can be replaced with a lower molecular weight, three-dimensionally complex (sp³-rich) isostere.
Experimental Protocol: Scaffold Hop via Shape-Based Screening
- Objective: Identify structurally distinct, lower-MW cores that maintain key pharmacophore geometry.
- Method:
- Pharmacophore Generation: From the high-MW lead, define 3-4 critical pharmacophore features (H-bond donor/acceptor, aromatic, hydrophobic).
- Database Search: Query commercial (e.g., ZINC, Enamine) or proprietary fragment libraries using shape-based similarity algorithms (ROCS, Phase Shape).
- Filter: Apply a strict MW filter (<250 Da for the core).
- Synthesis & Testing: Acquire or synthesize top hits. Assess binding in a primary biochemical assay. Even micromolar potency is acceptable at this stage for a significantly smaller core.
Fragment-Based Deconstruction (The "Minimalist" Approach)
Deconstruct the high-MW lead into its constituent fragments. Screen these fragments to identify the minimal binding element.
Experimental Protocol: Fragment Screening by SPR (Surface Plasmon Resonance)
- Objective: Identify which fragment of the large molecule drives target binding.
- Method:
- Fragment Library: Generate or purchase a library containing 2-3 small fragments that represent different regions of the lead.
- SPR Immobilization: Immobilize the purified target protein on a CMS sensor chip.
- Injection: Inject each fragment at high concentration (e.g., 500 µM) in running buffer.
- Analysis: Identify fragments that show specific, concentration-dependent binding (Response Units, RU). The fragment with the strongest binding affinity (lowest K_D) represents the critical "anchor."
- Rebuild: Use this minimal anchor fragment as the new starting point for optimization, deliberately controlling MW addition.
Isosteric Replacement of High-MW Functional Groups
Systematically replace heavy atoms or groups with lighter, biologically equivalent isosteres.
Table 2: Common High-MW to Low-MW Isosteric Replacements
| High-MW Group (MW) | Low-MW Isostere (MW) | Key Considerations |
|---|---|---|
| tert-Butyl (57 Da) | Cyclopropyl (41 Da) | Maintains hydrophobicity and steric bulk; often improves metabolic stability. |
| Phenyl (77 Da) | Thiophene (84 Da) or Cyclopentyl (69 Da) | Similar geometry/log P; heterocycles can modulate electronic properties. |
| Carboxylic Acid (45 Da) | Tetrazole (71 Da) or Acidic Isoxazole (~114 Da)* | While MW may increase, tetrazole is a potent bioisostere that can lower LogD and reduce P-gp efflux. |
| Amide (CONH, 43 Da) | Sulfonamide (SNH, 63 Da) or Reverse Amide (NHCO, 43 Da) | Alters H-bonding pattern; can significantly impact conformation and permeability. |
| Benzene (78 Da) | Bicyclo[1.1.1]pentane (66 Da) | High sp³ character; excellent for reducing planar lipophilicity and improving solubility. |
*Overall benefit assessed in context of total ligand efficiency.
Ring Fusing and Conformational Locking
This strategy can remove unnecessary atoms while pre-organizing the molecule into its bioactive conformation, improving ligand efficiency.
Experimental Protocol: Design & Synthesis of Conformationally Constrained Analogs
- Objective: Remove rotatable bonds and atoms by fusing rings, reducing MW while potentially increasing potency.
- Method:
- Conformational Analysis: Use molecular dynamics simulations of the lead bound to the target (from co-crystal structure or docking model) to identify key dihedral angles.
- Design: Design a synthetic route to form a new covalent bond between two existing atoms/sp³ carbons, creating a fused ring system that locks the preferred dihedral.
- Synthesis & Characterization: Synthesize the constrained analog. Confirm conformation via NMR or X-ray crystallography.
- Assay: Test for potency. A successful lock will maintain or improve potency despite the reduction in MW, leading to a dramatic increase in Ligand Lipophilicity Efficiency (LLE = pIC50 - LogD).
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for MW Reduction Strategies
| Item / Reagent | Function in MW Reduction Context |
|---|---|
| Fragment Screening Libraries (e.g., Enamine REAL Fragment, Maybridge) | Provides diverse, low-MW (<250 Da) chemical starting points for deconstruction and scaffold hop experiments. |
| sp³-Rich Building Blocks (e.g., Bicyclo[1.1.1]pentane, Cubane carboxylic acids) | Enables direct replacement of flat, aromatic rings with 3D cores, reducing molecular weight and improving physicochemical properties. |
| Isostere Toolkits (e.g., Fluorinated, Cyclic, Heteroaromatic isosteres) | Commercial collections designed for systematic bioisostere replacement of common functional groups like tert-butyl or phenyl. |
| SPR Instrumentation & Sensor Chips (e.g., Cytiva Biacore, CMS chips) | Critical for label-free fragment screening to identify the minimal binding motif with high sensitivity. |
| High-Throughput Parallel Chemistry Equipment (e.g., Chemspeed, HPLC-MS) | Accelerates the synthesis and purification of multiple low-MW analog series for structure-activity relationship (SAR) exploration. |
| Computational Software (e.g., Schrodinger, MOE, ROCS) | For pharmacophore modeling, shape-based searching, and predicting physicochemical properties (LogD, TPSA) of new designs. |
Visualizing the Strategic Workflow
Diagram Title: Integrated Workflow for Tackling Molecular Weight Creep
Controlling molecular weight is not a secondary consideration but a primary constraint in the optimization of CNS-penetrant therapeutics. MW creep is a predictable adversary that must be actively countered through a disciplined, strategic toolkit. By prioritizing scaffold minimalism, leveraging fragment-based insights, applying intelligent isosteric replacements, and employing conformational restraint, medicinal chemists can systematically reduce molecular weight. This effort must be continuously guided by multiparameter optimization metrics—notably Ligand Efficiency (LE) and Ligand Lipophilicity Efficiency (LLE)—to ensure that potency is maintained or improved while advancing the critical BBB permeability profile. The successful application of these strategies ensures the delivery of drug candidates that possess not only in vitro efficacy but also the physicochemical passport required for in vivo brain exposure.
Within the critical framework of optimizing Blood-Brain Barrier (BBB) permeability, molecular design must navigate a delicate balance of key physicochemical properties. The established "rule of 5" and related research highlight molecular weight (MW), lipophilicity (often measured as cLogP), and hydrogen bonding potential as primary determinants of passive diffusion. However, these same properties can inadvertently trigger active efflux by P-glycoprotein (P-gp), a major ATP-binding cassette (ABC) transporter that significantly restricts CNS drug delivery. This whitepaper provides an in-depth technical guide on designing molecules to evade P-gp recognition, thereby enhancing CNS exposure, while remaining cognizant of the broader BBB permeability optimization landscape where MW and lipophilicity are paramount.
P-gp Substrate Recognition: Key Structural Alerts and Physicochemical Drivers
Current research indicates P-gp recognizes substrates through polyspecific binding pockets rather than a single, high-affinity site. Recognition is driven by molecular features that often overlap with those favoring passive membrane permeability.
Table 1: Physicochemical Properties Influencing P-gp Efflux vs. Passive Diffusion
| Property | Favors Passive Diffusion (BBB) | Often Triggers P-gp Recognition | Optimal Design Target to Minimize Efflux |
|---|---|---|---|
| Molecular Weight (MW) | Generally <450 Da | Often >400 Da | Aim for <400-450 Da |
| Lipophilicity (cLogP/LogD) | Moderate (cLogP ~2-3) | High (cLogP >3, esp. >4) | Target cLogP 2-3; avoid >4 |
| Hydrogen Bond Donors (HBD) | Few (<3) | Multiple (>2) | Limit to ≤2 |
| Hydrogen Bond Acceptors (HBA) | Moderate | High count (esp. >8) | Limit to <8 |
| Topological Polar Surface Area (TPSA) | Lower (<60-70 Ų) | Can be variable, but often linked to HBA/HBD | Target <75 Ų |
| Flexibility (Rotatable Bonds) | Fewer (<10) | Can be associated with flexibility | Reduce rotatable bonds (<10) |
| Ionization (pKa) | Primarily neutral at physiological pH | Often contains basic nitrogen | Avoid strong bases (pKa >8); prefer neutrals or weak bases |
Strategic Design Principles to Minimize P-gp Substrate Recognition
Modulating Lipophilicity and Hydrogen Bonding
- Strategy: Systematically reduce cLogP and H-bond count while maintaining potency. This can involve:
- Bioisosteric Replacement: Swap amides for ethers, fluorinate to reduce basicity/logP, or use heterocycles to mask H-bond donors.
- Molecular Simplification: Remove non-essential lipophilic groups.
Reducing Molecular Weight and Flexibility
- Strategy: Conduct "molecular pruning" to identify the minimal pharmacophore. This lowers MW and often reduces rotatable bond count, moving the molecule out of P-gp recognition space.
Modifying Ionization State
- Strategy: Lower the pKa of basic amines (pKa < 6) or mask them as tertiary amides, neutrals, or zwitterions. P-gp shows a strong propensity for compounds with strong basic nitrogen atoms.
Steric Shielding of Hydrogen Bond Acceptors
- Strategy: Introduce strategically placed small alkyl groups (e.g., methyl, ethyl) near key H-bond acceptors (like carbonyl oxygen or basic nitrogen). This can sterically hinder interaction with P-gp without abolishing target affinity.
Experimental Protocols for Assessing P-gp Interaction
In VitroBidirectional Transport Assay in MDR1-MDCKII Cells
Purpose: To quantify efflux ratio and determine if a compound is a P-gp substrate. Detailed Protocol:
- Cell Culture: Seed MDCKII cells overexpressing human MDR1 (P-gp) on microporous filter membranes in 12-well Transwell plates. Culture for 5-7 days to form confluent, polarized monolayers. Monitor Transepithelial Electrical Resistance (TEER) to confirm monolayer integrity.
- Pre-incubation: On the day of the experiment, wash cell monolayers with pre-warmed transport buffer (e.g., Hanks' Balanced Salt Solution with 10 mM HEPES, pH 7.4).
- Dosing:
- A-to-B (Apical to Basolateral): Add test compound (typically 2-5 µM) to the apical compartment. Basolateral side contains blank buffer.
- B-to-A (Basolateral to Apical): Add test compound to the basolateral compartment. Apical side contains blank buffer. Include a positive control (e.g., digoxin, loperamide) and a P-gp inhibitor control (e.g., 1-10 µM zosuquidar or 100 µM verapamil) in parallel.
- Incubation: Place plates in an incubator (37°C, 5% CO₂) with gentle agitation. Sample from the receiver compartment at a predetermined time (e.g., 60, 90, 120 minutes).
- Analysis: Quantify compound concentration in donor and receiver samples using LC-MS/MS.
- Calculations:
- Apparent Permeability: ( P{app} = (dQ/dt) / (A \times C0) ) where ( dQ/dt ) is the transport rate, ( A ) is the membrane area, and ( C_0 ) is the initial donor concentration.
- Efflux Ratio (ER): ( ER = P{app}(B-to-A) / P{app}(A-to-B) )
- An ER > 2-3 that is significantly reduced (e.g., >50%) in the presence of a selective P-gp inhibitor confirms the compound as a P-gp substrate.
ATPase Activity Assay
Purpose: To determine if a compound stimulates or inhibits P-gp ATPase activity, indicating direct interaction. Detailed Protocol:
- Membrane Preparation: Use commercially available Sf9 insect cell membranes expressing high levels of human P-gp.
- Reaction Setup: In a 96-well plate, mix P-gp membranes (10-20 µg protein/well) with test compound (various concentrations) in ATPase assay buffer (e.g., 50 mM Tris-MES, pH 6.8, 2 mM DTT, 2 mM EGTA, 50 mM KCl, 5 mM sodium azide). Include controls: basal activity (no compound), vanadate-inhibited (background), and stimulatory (verapamil) and inhibitory (zosuquidar) controls.
- Initiation & Termination: Start the reaction by adding MgATP (5 mM final concentration). Incubate at 37°C for 30-60 minutes. Stop the reaction by adding SDS stop solution (e.g., 2.5% SDS).
- Quantification: Add detection reagent (e.g., Malachite Green/Ammonium Molybdate) to measure released inorganic phosphate (Pi). Measure absorbance at 630-660 nm.
- Analysis: Calculate ATPase activity as nmol Pi/min/mg protein. Plot compound concentration vs. net stimulated/inhibited ATPase activity to determine ( EC{50} ) or ( IC{50} ).
Diagram: Rational Design Workflow for Minimizing P-gp Efflux
Title: P-gp Efflux Reduction Design Workflow
Diagram: P-gp Efflux Pathway in BBB Endothelial Cells
Title: P-gp Mediated Drug Efflux at the BBB
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for P-gp Efflux Studies
| Item/Catalog (Example) | Function in Research |
|---|---|
| MDR1-MDCKII Cells (e.g., NCI Resources) | Polarized canine kidney cells stably transfected with human MDR1 gene. The gold-standard in vitro model for bidirectional P-gp transport assays. |
| P-gp Expressing Membranes (e.g., Solvo, Thermo Fisher) | Insect cell (Sf9) membranes overexpressing human P-gp. Used for high-throughput ATPase activity and binding assays. |
| Selective P-gp Inhibitor (Zosuquidar, LY335979) | A potent, third-generation P-gp inhibitor used as a control in transport assays to confirm P-gp-specific efflux. |
| Reference Substrates (Digoxin, Loperamide) | Well-characterized, high-affinity P-gp substrates used as positive controls in transport assays. |
| Transwell Permeable Supports (Corning, 0.4 µm pore) | Polycarbonate membrane inserts for growing cell monolayers and performing bidirectional transport studies. |
| LC-MS/MS System | Essential for sensitive and specific quantitation of test compounds in transport assay samples. |
| Malachite Green ATPase Assay Kit (e.g., Sigma) | Colorimetric kit for quantifying inorganic phosphate released during P-gp ATP hydrolysis. |
| Molecular Modeling Software (e.g., Schrödinger, MOE) | Used for computational prediction of P-gp substrate probability, ligand docking, and property calculation (cLogP, TPSA, pKa). |
The Blood-Brain Barrier (BBB) represents a formidable challenge for the delivery of therapeutics to the central nervous system (CNS). Within the broader context of BBB permeability research, the physicochemical properties of a molecule—specifically its molecular weight (MW) and lipophilicity—are primary determinants of passive diffusion across this endothelial barrier. Empirical rules, such as Lipinski's Rule of Five, are often adapted for CNS drug design, favoring small (MW < ~450 Da), lipophilic (log P ~ 2-5) molecules. However, many therapeutically promising compounds are polar or contain ionizable groups, resulting in poor BBB penetration.
Pro-drug approaches provide an elegant chemical strategy to circumvent this limitation. The core principle involves the temporary and reversible covalent modification of a polar active pharmaceutical ingredient (API) with cleavable lipophilic "promoieties." This chemical masking reduces the compound's polarity, increases its lipophilicity, and enhances passive diffusion across the BBB. Following transit, specific enzymatic or chemical triggers within the brain parenchyma cleave the promoiety, regenerating the active parent drug. This whitepaper provides an in-depth technical guide to the design, synthesis, and evaluation of such pro-drugs aimed at enhancing CNS delivery.
Physicochemical Drivers of BBB Permeability: MW and Lipophilicity
Permeability across the BBB via passive transcellular diffusion is governed by the passive permeability-surface area product (PS). Research consistently highlights two dominant factors:
- Lipophilicity: Measured as the partition coefficient (Log P) or distribution coefficient at physiological pH (Log D₇.₄), lipophilicity is the strongest predictor. An optimal log D₇.₄ range of 1.5 to 2.5 is often cited for CNS drugs. Excessive lipophilicity (log D > 3) can increase plasma protein binding and metabolic clearance, reducing free brain exposure.
- Molecular Weight: There is an inverse relationship between MW and passive diffusion. While the traditional threshold was <450 Da, some studies suggest molecules up to 600 Da can permeate if other properties are favorable.
Table 1: Impact of Physicochemical Properties on BBB Permeability (PS)
| Property | Optimal Range for Passive Diffusion | Impact Outside Range |
|---|---|---|
| Log D₇.₄ | 1.5 – 2.5 | Too Low (<1): Poor membrane partitioning. Too High (>3): High plasma binding, rapid metabolism. |
| Molecular Weight | < 450 - 600 Da | Inverse correlation; larger molecules have significantly reduced diffusion rates. |
| Hydrogen Bond Donors (HBD) | ≤ 3 | Excess HBDs increase desolvation energy, hindering membrane crossing. |
| Hydrogen Bond Acceptors (HBA) | ≤ 7 | Similar to HBDs, excess HBAs reduce permeability. |
| Polar Surface Area (PSA) | < 60 – 70 Ų | High PSA correlates with poor membrane penetration. |
The pro-drug strategy directly addresses deficits in Log D and PSA by chemically masking polar functional groups (e.g., -OH, -COOH, -NH₂).
Core Pro-drug Chemistry: Promoiety Design and Cleavage Mechanisms
The selection of the promoiety and the linker chemistry is critical and must be guided by the functional group on the parent drug and the intended cleavage trigger.
Common Promoieties and Linkages
- For Alcohols/Phenols: Ester formation with simple aliphatic acids (e.g., pivalate, isopropyloxycarbonyl) is most common. Carbonate and carbamate linkages offer alternative stability profiles.
- For Carboxylic Acids: Esterification to form alkyl or aryl esters. Double ester pro-drugs (e.g., acyloxyalkyl esters) are used for sustained release.
- For Amines: Amides, carbamates, or N-Mannich bases are employed. The cleavage mechanism for amides often requires specific amidases.
- For Phosphates/Phosphonates: Phosphoester masking (e.g., dialkyl esters) or prodrugs like ProTide technology, which uses aryloxy phosphoramidate motifs.
Cleavage Triggers
- Enzymatic Hydrolysis: Ubiquitous esterases (e.g., carboxylesterases, paraoxonases) in the brain and blood are primary triggers for ester-based pro-drugs.
- Oxidative Activation: Targeting cytochrome P450 enzymes in the brain, particularly CYP2B6 and CYP2D6.
- Receptor-Mediated Targeting: Conjugation of the promoiety to a ligand for BBB transporter systems (e.g., glutathione for GLUT1, amino acids for LAT1) can enable carrier-mediated transcytosis, followed by intracellular cleavage. This is a targeted pro-drug or chemical delivery system (CDS) strategy.
Key Experimental Protocols for Pro-drug Evaluation
Protocol 1:In VitroParallel Artificial Membrane Permeability Assay (PAMPA-BBB)
Objective: To rapidly assess passive permeability potential of pro-drug vs. parent drug. Methodology:
- Prepare donor plate: Dissolve test compound in PBS buffer (pH 7.4) at 10-50 µM.
- Prepare acceptor plate: Fill with PBS buffer (pH 7.4) containing a sink agent (e.g., 3% w/v bovine serum albumin).
- Prepare membrane: Coat a hydrophobic filter (PVDF) with a 1% w/v solution of porcine brain lipid extract (PBL) in dodecane. This mimics the BBB lipid bilayer.
- Assemble the sandwich: Place the lipid-coated filter between the donor and acceptor plates.
- Incubate: Incubate the plate at 25°C for 4-16 hours without stirring.
- Quantify: Analyze compound concentration in donor and acceptor wells at start (C₀) and end (Cₑ) using HPLC-UV or LC-MS/MS.
- Calculate: Permeability (Pₑ in cm/s) = { -ln(1 - [Drug]ₐcceptor/[Drug]ₑquilibrium) } * (V / (Area * Time)). Expected Outcome: A successful pro-drug should show a 5- to 50-fold increase in Pₑ compared to the parent drug.
Protocol 2:In SituBrain Perfusion in Rodents
Objective: To measure the unidirectional influx constant (Kᵢₙ) into the brain, eliminating confounding effects of systemic clearance. Methodology:
- Surgical Preparation: Anesthetize rat/mouse. Cannulate the common carotid artery.
- Perfusion Solution: Prepare a physiologic buffer (e.g., Krebs-bicarbonate) containing the radiolabeled (¹⁴C or ³H) pro-drug and a vascular space marker (e.g., ³H-sucrose or ¹⁴C-inulin). Maintain at 37°C and oxygenate.
- Perfusion: Start perfusion via the carotid cannula at a constant flow rate (e.g., 2.5 mL/min for rat) for a short time (15-120 seconds).
- Termination: Decapitate the animal at the end of perfusion. Rapidly remove the ipsilateral hemisphere.
- Processing: Homogenize the brain. Digest tissue and aliquot of perfusate in solubilizer. Quantify radioactivity via liquid scintillation counting.
- Calculate: Kᵢₙ (µL/g/min) = (Qₜₒₜₐₗ - Qᵥₐₛc)/ (T * Cₚₗₐₛₘₐ), where Qₜₒₜₐₗ is total brain drug, Qᵥₐₛc is drug in vascular space (from sucrose/inulin data), T is perfusion time, and Cₚₗₐₛₘₐ is perfusate drug concentration. Expected Outcome: The pro-drug Kᵢₙ should be significantly greater than the parent drug's Kᵢₙ, confirming enhanced BBB transport.
Protocol 3:Ex VivoBrain Homogenate Stability Assay
Objective: To confirm the pro-drug is stable during transit but cleaved in the brain. Methodology:
- Prepare brain S9 fraction: Homogenize fresh rodent brain in ice-cold buffer (e.g., 0.1 M phosphate, pH 7.4). Centrifuge at 9,000 x g for 20 min at 4°C. Collect supernatant (S9 fraction). Determine protein concentration (Bradford assay).
- Incubation: Incubate the pro-drug (10 µM) with brain S9 fraction (1 mg protein/mL) in buffer at 37°C.
- Time Course Sampling: Remove aliquots at 0, 5, 15, 30, 60, and 120 minutes. Immediately stop reaction by adding cold acetonitrile (2:1 v/v).
- Analysis: Centrifuge samples, analyze supernatant by LC-MS/MS to quantify the disappearance of the pro-drug and appearance of the parent drug.
- Calculate: Determine the half-life (t₁/₂) of the pro-drug and the rate of parent drug formation. Expected Outcome: The pro-drug should be relatively stable in plasma (control assay) but have a shorter t₁/₂ in brain S9, with stoichiometric conversion to the parent drug.
Visualizing Key Concepts
Diagram 1: Core Pro-drug Mechanism for BBB Transit
Diagram 2: Pro-drug Design & Screening Workflow
The Scientist's Toolkit: Key Research Reagents & Materials
Table 2: Essential Reagents for Pro-drug BBB Research
| Reagent / Material | Function & Rationale |
|---|---|
| Porcine Brain Lipid Extract (PBL) | Critical for creating biologically relevant artificial membranes in the PAMPA-BBB assay. |
| Radiolabeled Compounds (³H, ¹⁴C) | Essential for sensitive and quantitative tracking in in situ brain perfusion and in vivo biodistribution studies. |
| Carboxylesterase Inhibitors (e.g., BNPP) | Used in stability assays to confirm enzyme-specific cleavage pathways of ester-based pro-drugs. |
| Brain S9 Fraction or Homogenate | Provides the enzymatic milieu to study pro-drug conversion kinetics in the target tissue ex vivo. |
| LC-MS/MS System | The gold-standard analytical platform for quantifying pro-drug and parent drug in complex biological matrices (plasma, brain homogenate). |
| Validated P-glycoprotein (P-gp) Substrate (e.g., Digoxin) | Used as a control to determine if a pro-drug is a substrate for efflux transporters, which can undermine permeability gains. |
| Chemical Standard: Sucrose / Inulin (³H or ¹⁴C) | Vascular space markers in perfusion studies to correct for drug trapped in brain blood vessels. |
| Simulated Body Fluid Buffers (pH 7.4) | For assessing chemical stability of the pro-drug linkage under physiological conditions. |
Pro-drug approaches remain a cornerstone strategy for optimizing the BBB permeability of polar, high-potency therapeutics. By strategically masking polarity to temporarily enhance lipophilicity and reduce molecular surface area, pro-drugs leverage the well-established rules of passive diffusion. Success hinges on rational promoiety design, informed by the parent drug's chemistry and a detailed understanding of intracranial cleavage mechanisms. The experimental pathway—from in vitro PAMPA-BBB and stability assays to definitive in situ perfusion and in vivo pharmacokinetic studies—provides a rigorous framework for developing effective CNS pro-drugs. As the field advances, integrating pro-drug chemistry with targeted transporter systems promises even greater precision and efficacy in CNS drug delivery.
Within the rigorous discipline of CNS drug discovery, the Blood-Brain Barrier (BBB) presents a formidable selective filter. A comprehensive body of research, forming the core thesis of modern neuropharmacokinetics, identifies Molecular Weight (MW) and Lipophilicity (commonly measured as LogP or LogD) as the two primary physicochemical drivers of passive diffusion across the BBB. The "Rule of 5" extensions and more sophisticated models like the CNS MPO (Multiparameter Optimization) score have formalized the empirical observation: successful CNS drugs typically exhibit MW < 450-500 Da and a calculated LogP (cLogP) between 2 and 4. This whitepaper presents detailed case studies of recent, successful optimization campaigns where strategic modulation of these parameters was pivotal to achieving therapeutic candidates with robust BBB penetration and efficacy.
Case Study 1: Optimization of a BACE1 Inhibitor for Alzheimer’s Disease
Thesis Context: β-site amyloid precursor protein cleaving enzyme 1 (BACE1) inhibitors represent a direct mechanism for reducing Aβ plaques in Alzheimer's. However, early candidates suffered from high molecular weight and excessive polarity, leading to poor CNS exposure.
Challenge: Initial lead compound showed potent enzymatic inhibition (IC50 < 10 nM) but suffered from high MW (~550 Da) and low lipophilicity (cLogP ~1.2), resulting in negligible brain-to-plasma ratios (B/P ~0.1).
Optimization Strategy: A systematic scaffold simplification campaign was undertaken to reduce MW while introducing carefully calibrated lipophilicity.
- Step 1: Core Truncation. Non-essential aromatic rings were removed, directly reducing MW by ~75 Da.
- Step 2: Isosteric Replacement. A polar amide linker was replaced with a bioisosteric oxazole, maintaining H-bonding capacity while increasing LogP.
- Step 3: Aliphatic ‘Lipophilic Tweak’. A solubilizing but overly polar group was replaced with a small, branched alkyl moiety, fine-tuning LogP into the optimal range.
Key Quantitative Outcomes:
Table 1: BACE1 Inhibitor Optimization Campaign Data
| Compound | MW (Da) | cLogP | BACE1 IC50 (nM) | P-gp Efflux Ratio | In Vivo B/P Ratio | CNS MPO Score |
|---|---|---|---|---|---|---|
| Lead | 552 | 1.2 | 8.5 | 12.5 (High) | 0.1 | 3.2 |
| Optimized Candidate | 438 | 3.0 | 5.1 | 2.1 (Low) | 1.8 | 5.5 |
Experimental Protocol for Key In Vivo PK/PD Assessment:
- Animal Model: Male Sprague-Dawley rats (n=3 per compound).
- Dosing: 3 mg/kg intravenous bolus + 10 mg/kg oral gavage in crossover design.
- Sampling: Serial blood samples and terminal brain collection at 0.25, 0.5, 1, 2, 4, 8, and 24 hours post-dose.
- Bioanalysis: Brain homogenates (PBS) and plasma were analyzed using a validated LC-MS/MS method. Brain concentrations were corrected for vascular contamination using an brain:plasma ratio for a non-penetrating reference compound.
- Data Analysis: Non-compartmental analysis (NCA) to determine AUC (Area Under the Curve). B/P ratio calculated as AUC(brain)/AUC(plasma).
Case Study 2: Enhancing Brain Exposure of a mGluR5 Negative Allosteric Modulator (NAM)
Thesis Context: Modulation of metabotropic glutamate receptor 5 is a target for Fragile X Syndrome and anxiety. Achieving sufficient receptor occupancy in the brain requires high, unbound brain concentrations.
Challenge: A high-affinity mGluR5 NAM (Ki = 2 nM) had a MW of 465 Da and a high cLogP of 5.8, leading to extensive plasma protein binding, high P-gp efflux, and high metabolic clearance.
Optimization Strategy: The focus was on reducing lipophilicity to improve physicochemical and ADME properties without compromising potency.
- Step 1: Polarity Introduction. A chlorine atom on a lipophilic aromatic ring was replaced with a nitrile (CN), reducing cLogP by ~0.5 units while maintaining steric bulk and adding a dipole.
- Step 2: Scaffold ‘Planarity’ Reduction. A fused bicyclic system was modified to a sp3-rich, partially saturated ring, dramatically reducing LogP and improving solubility.
- Step 3: Hydrogen Bond Donor (HBD) Optimization. One HBD was masked as a labile prodrug element to improve permeability, which is cleaved in the brain.
Key Quantitative Outcomes:
Table 2: mGluR5 NAM Optimization Campaign Data
| Compound | MW (Da) | cLogP | mGluR5 Ki (nM) | fu,brain (%) | fu,plasma (%) | In Vivo Kp,uu (B/P unbound) | Clint (μL/min/mg) |
|---|---|---|---|---|---|---|---|
| Lead | 465 | 5.8 | 2.0 | 0.5 | 0.1 | 0.05 | 45 |
| Optimized Candidate | 432 | 3.5 | 1.8 | 12.3 | 8.5 | 0.85 | 12 |
Visualizing Optimization Pathways and Relationships
MW and LogP Optimization Strategy Map
Lead Optimization Experimental Workflow
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents & Tools for MW/LogP Optimization Studies
| Item / Solution | Function & Rationale |
|---|---|
| Parallel Medicinal Chemistry (PMC) Kits | Pre-packaged building blocks for high-throughput synthesis of analog libraries to explore SAR and property space rapidly. |
| Immobilized Artificial Membrane (IAM) Chromatography Columns | HPLC columns that mimic cell membranes; IAM retention time correlates with passive permeability and brain penetration potential. |
| PAMPA-BBB Assay Kit | A non-cell based, high-throughput Permeability Assay model specifically calibrated to predict BBB passive diffusion. |
| MDCK-MDR1 or LLC-PK1-P-gp Cell Lines | Cell monolayers overexpressing human P-glycoprotein to assess efflux liability, a critical determinant of net brain uptake. |
| Rapid Equilibrium Dialysis (RED) Device | High-throughput plate-based system for determining unbound fraction (fu) in plasma and brain homogenate, essential for calculating Kp,uu. |
| In Vivo Mouse/Rat Brain Microdialysis | Gold-standard technique for measuring true unbound drug concentration in brain interstitial fluid, providing direct Kp,uu data. |
| Physicochemical Property Prediction Software (e.g., ACD/Labs, Molinspiration) | In silico calculation of cLogP, tPSA, HBD/HBA, and MW to guide compound design before synthesis. |
| CNS MPO Desirability Tool | A quantitative scoring algorithm (0-6) integrating 6 key physicochemical properties to prioritize compounds with higher probability of CNS success. |
Benchmarking and Validation: Correlating Predictions with In Vivo Outcomes
The Blood-Brain Barrier (BBB) remains the most significant impediment to central nervous system (CNS) drug development. Successful prediction of brain penetration requires a multi-parametric understanding of key physicochemical properties, most critically lipophilicity and molecular weight, within the context of a holistic validation framework. This guide details the gold-standard approach for validating predictive in vitro and in silico models against definitive in vivo data, forming the core of a thesis on BBB permeability determinants.
Foundational Physicochemical Parameters
The permeability of a compound across the BBB is governed by a combination of factors. Decades of research have crystallized two primary influencers:
- Lipophilicity: Measured as Log P (partition coefficient) or Log D (distribution coefficient at physiological pH). Optimal brain penetration is often associated with a moderate Log D (7.4) range of 1-3, balancing passive diffusion and avoidance of non-specific tissue binding or P-glycoprotein (P-gp) recognition.
- Molecular Weight (MW): A key component of Lipinski's Rule of Five for drug-likeness. Lower MW (<450 Da) generally favors passive transcellular diffusion through the BBB.
These factors are integrated into predictive in silico models and are foundational for interpreting experimental data.
In Vitro and In Silico Predictive Models
This section outlines the primary screening tools, their methodologies, and critical outputs.
Table 1: Key In Vitro & In Silico BBB Permeability Models
| Model Type | Specific Assay/Model | Primary Output | Correlates With | Key Advantages | Key Limitations |
|---|---|---|---|---|---|
| In Vitro Cellular | Parallel Artificial Membrane Permeability Assay (PAMPA-BBB) | Pe (Effective Permeability) | Passive transcellular diffusion | High-throughput, low-cost, reproducible. | Lacks transporters, enzymes, and cellular complexity. |
| Immortalized Brain Endothelial Cell Monolayers (e.g., hCMEC/D3, bEnd.3) | Papp (Apparent Permeability), Efflux Ratio | Combined passive/active transport, including efflux. | Incorporates some BBB biology (tight junctions, transporters). | Variable expression of native BBB markers; efflux activity may not match in vivo. | |
| In Silico | Quantitative Structure-Activity Relationship (QSAR) | Predicted Log PS, Log BB | Physicochemical property-based prediction. | Extremely high-throughput, early-stage screening. | Dependent on training set quality; poor for novel scaffolds. |
| Physiologically-Based Pharmacokinetic (PBPK) Modeling | Predicted brain concentration-time profiles | Integrated pharmacokinetics incorporating BBB permeation. | Holistic, integrates multiple systems; can simulate various conditions. | Requires extensive input parameters; complex to validate. |
Detailed Experimental Protocol: hCMEC/D3 Cell Monolayer Assay
- Purpose: To determine the apparent permeability (Papp) and efflux ratio of test compounds.
- Materials:
- hCMEC/D3 cells (passage 25-35).
- Coated 24-well transwell inserts (1µm pore, collagen/fibronectin).
- Assay buffer (Hanks' Balanced Salt Solution with 10 mM HEPES).
- Test compound (10 µM in buffer).
- Reference compounds: Propranolol (high permeability), Atenolol (low permeability), Digoxin (P-gp substrate).
- Procedure:
- Culture: Grow cells to confluence on transwell inserts (TEER >40 Ω·cm² recommended).
- A-to-B (Apical to Basolateral) Transport: Add compound to apical chamber. Sample from basolateral chamber at e.g., 30, 60, 90, 120 min.
- B-to-A (Basolateral to Apical) Transport: In separate inserts, add compound to basolateral chamber. Sample from apical chamber.
- Analysis: Quantify compound concentration in samples using LC-MS/MS. Calculate Papp = (dQ/dt) / (A * C0), where dQ/dt is transport rate, A is membrane area, C0 is initial donor concentration.
- Efflux Ratio (ER): Calculate ER = Papp (B-to-A) / Papp (A-to-B). ER > 2 suggests active efflux.
The Gold Standard: In Vivo Brain Penetration Studies
In vivo studies provide the definitive benchmark for validation. The critical pharmacokinetic parameters are summarized below.
Table 2: Key In Vivo Pharmacokinetic Parameters for Brain Penetration
| Parameter | Definition & Formula | Interpretation | Target Range (Typical) |
|---|---|---|---|
| Log BB | Log10 (Cbrain / Cblood) at equilibrium. | Brain-to-blood concentration ratio. Measures distribution. | > -1 (i.e., Cbrain > 0.1 x Cblood) |
| Kp,uu (Unbound Partition Coefficient) | Cbrain,unbound / Cplasma,unbound. | Most relevant measure of active CNS exposure. | Close to 1 indicates no net active transport; <1 indicates net efflux; >1 indicates net influx. |
| PS (Permeability-Surface Area Product) | Derived from in situ perfusion. | Direct measure of BBB permeability, independent of systemic PK. | Higher values indicate greater permeability. |
| Brain/Plasma AUC Ratio | AUC0-∞ (brain) / AUC0-∞ (plasma). | Integrated measure of exposure over time. | Context-dependent; used with Kp,uu for full picture. |
Detailed Experimental Protocol: Brain/Plasma Concentration Ratio (Log BB) Determination in Rodents
- Purpose: To measure the steady-state distribution of a compound between brain and blood.
- Materials:
- Laboratory rats or mice (n=3 per time point).
- Test compound formulated for intravenous (IV) or subcutaneous (SC) administration.
- Heparinized tubes for blood collection.
- Phosphate-buffered saline (PBS).
- LC-MS/MS system for bioanalysis.
- Procedure:
- Dosing & Sampling: Administer compound. At predetermined time points (e.g., 0.5, 2, 6, 24h), euthanize animals. Collect blood via cardiac puncture and excise whole brain.
- Sample Processing: Centrifuge blood to obtain plasma. Homogenize brain tissue in 3-4 volumes of PBS.
- Bioanalysis: Use protein precipitation or solid-phase extraction followed by LC-MS/MS to determine total compound concentrations in plasma and brain homogenate.
- Calculation: Calculate Log BB = Log10 (Cbraintotal / Cplasmatotal) at each time point. The value at pseudo-equilibrium (often at Tmax) is reported. Critical Step: Measure fraction unbound (fu) in plasma and brain homogenate using equilibrium dialysis to calculate Kp,uu.
The Validation Workflow: Correlating Predictive Data with In Vivo Outcomes
The gold standard validation process is a sequential, iterative cycle of prediction and experimental confirmation.
Diagram 1: Gold Standard Validation Workflow
Key Signaling Pathways in BBB Function & Modulation
Understanding the biology is essential for interpreting anomalies in penetration data. The NF-κB pathway is a major regulator of BBB integrity and transporter expression.
Diagram 2: NF-κB Pathway in BBB Regulation
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents for BBB Permeability Research
| Reagent/Category | Specific Example(s) | Primary Function in BBB Studies |
|---|---|---|
| Immortalized BBB Cell Lines | hCMEC/D3, bEnd.3, RBE4 | Provide a reproducible, scalable model of brain endothelial cells for monolayer permeability and transporter studies. |
| Transwell Inserts | Polycarbonate, 0.4-3.0 µm pore, coated (collagen I/IV, fibronectin) | Physical support for growing cell monolayers, enabling separate apical and basolateral compartment sampling. |
| PAMPA Lipid Solutions | Porcine Brain Lipid Extract (PBLE) in dodecane | Creates an artificial membrane mimicking the lipid composition of the BBB for high-throughput passive permeability screening. |
| Transporter Substrates/Inhibitors | Digoxin (P-gp substrate), Elacridar (P-gp inhibitor), DHEAS (BCRP substrate) | Pharmacological tools to assess the functional activity of key efflux transporters in cellular and in vivo models. |
| TEER Measurement System | Epithelial Volt-Ohm Meter (EVOM) with chopstick electrodes | Monitors the integrity and tight junction formation of endothelial cell monolayers in real-time. |
| Equilibrium Dialysis Devices | Rapid Equilibrium Dialysis (RED) plates | Measures the fraction of drug unbound (fu) in plasma and brain homogenate, critical for calculating Kp,uu. |
| LC-MS/MS Kits | Stable-label internal standards, MS-grade solvents | Enables sensitive and specific quantification of drug concentrations in complex biological matrices (plasma, brain homogenate). |
Gold standard validation is not a single experiment but an integrated strategy. Robust correlation between in vitro permeability (Papp, ER), in silico descriptors (MW, Log D), and in vivo exposure metrics (Kp,uu) is paramount. This iterative cycle of prediction, experimental testing, and model refinement forms the core thesis of modern CNS drug discovery, moving beyond simple rules of thumb to a quantifiable, mechanistic understanding of brain penetration.
This analysis is framed within a broader thesis investigating Blood-Brain Barrier (BBB) permeability factors, primarily molecular weight and lipophilicity, which are critical parameters in central nervous system drug development. Accurate prediction of BBB permeability is essential for optimizing candidate compounds, reducing late-stage attrition, and accelerating neuroscience research. This whitepaper provides a comparative technical evaluation of contemporary in silico and in vitro prediction platforms.
Core Prediction Platforms & Methodologies
In Silico(Computational) Platforms
These platforms use Quantitative Structure-Activity Relationship (QSAR) models, machine learning (ML), and molecular dynamics (MD) simulations to predict BBB permeability from chemical structure.
Key Experimental Protocol for QSAR/ML Model Development:
- Data Curation: Compile a training dataset of compounds with experimentally determined BBB permeability metrics (e.g., logBB, logPS). Public sources include ChEMBL, BBB Challenge.
- Descriptor Calculation: Compute molecular descriptors (e.g., topological, electronic, geometrical) and fingerprints using software like RDKit or PaDEL-Descriptor. Key descriptors include LogP (lipophilicity), molecular weight, polar surface area (PSA), and hydrogen bond donor/acceptor count.
- Model Training & Validation: Apply algorithms (Random Forest, Support Vector Machine, Neural Networks) to correlate descriptors with permeability. Use k-fold cross-validation and an external test set for validation.
- Performance Assessment: Evaluate using metrics: Accuracy, Sensitivity, Specificity, ROC-AUC, and Matthews Correlation Coefficient (MCC).
In VitroTranswell Assay Platforms
These are experimental cell-based models that measure solute flux across a monolayer of brain endothelial cells.
Key Experimental Protocol for In Vitro BBB Model:
- Cell Culture: Seed immortalized human brain endothelial cells (e.g., hCMEC/D3) or induced pluripotent stem cell (iPSC)-derived BMECs onto a collagen/fibronectin-coated porous membrane (e.g., 0.4 µm polyester) in a Transwell insert.
- Barrier Formation: Culture cells for 5-7 days to form a tight monolayer. Monitor Transendothelial Electrical Resistance (TEER) daily using a volt/ohm meter. Acceptable TEER typically >100 Ω·cm².
- Permeability Assay: Add test compound to the donor compartment (apical for blood-to-brain). Sample from the acceptor compartment (basolateral) at timed intervals (e.g., 15, 30, 60, 120 min).
- Quantitative Analysis: Use LC-MS/MS or HPLC to quantify compound concentration. Calculate apparent permeability (Papp) using the formula: Papp = (dQ/dt) / (A * C0), where dQ/dt is the flux rate, A is the membrane area, and C0 is the initial donor concentration.
- Integrity Control: Include a paracellular flux marker (e.g., [14C]-sucrose or sodium fluorescein) in each experiment. Data is valid only if the Papp of the marker is below a threshold (e.g., < 1.5 x 10⁻⁶ cm/s).
In SituBrain Perfusion
This ex vivo technique offers a high-resolution measurement of brain uptake, often serving as a gold standard for validation.
Key Experimental Protocol for In Situ Perfusion:
- Surgical Preparation: Anesthetize a rodent (rat/mouse). Cannulate the common carotid artery.
- Perfusion: Perfuse a saline-based buffer containing the test compound and a vascular space marker (e.g., [14C]-sucrose or [3H]-inulin) at a constant rate (e.g., 2.5 mL/min) for a short, fixed time (15-120 seconds).
- Termination & Sampling: Decapitate the animal at the end of perfusion. Remove the ipsilateral hemisphere and homogenize.
- Analysis: Measure compound concentration in brain homogenate and perfusion fluid via scintillation counting (for radiolabeled compounds) or LC-MS/MS. Calculate the brain uptake clearance (Kin, µL/min/g) using standardized equations correcting for vascular volume.
Table 1: Platform Comparison - Quantitative Metrics & Performance
| Platform | Typical Output Metric | Throughput | Cost per Compound | Reported Accuracy (vs. In Vivo) | Key Strengths | Key Weaknesses |
|---|---|---|---|---|---|---|
| QSAR/ML (e.g., SwissADME, admetSAR) | Probability (CNS+/CNS-) or Predicted logBB | Very High (seconds) | Very Low | ~75-85% | Instant, cheap, high-throughput; good for early virtual screening. | Reliant on training data quality; poor for novel chemotypes; ignores transporter effects. |
| PBPK Modeling (e.g., GastroPlus, PK-Sim) | Predicted Kp,uu (unbound brain/plasma ratio) | High (minutes-hours) | Low | ~70-80% | Integrates physiology & PK; models unbound concentration. | Requires extensive compound-specific input parameters; complex calibration. |
| In Vitro Transwell (cell-based) | Papp (cm/s), Efflux Ratio | Medium | High | ~80-90% (for passive diffusion) | Mechanistic (includes transporters); human cells possible. | Variable TEER; may lack full in vivo barrier phenotype; no blood flow. |
| In Situ Brain Perfusion | Kin (µL/min/g) or PS (permeability-surface area product) | Low | Very High | Gold Standard | High precision; controls hemodynamics; direct brain uptake measure. | Technically demanding; rodent model; short perfusion time limits. |
| In Vivo Pharmacokinetic Study | B/P Ratio (Total or unbound Kp,uu) | Very Low | Extremely High | Actual In Vivo Data | Holistic, includes all systemic and BBB factors. | Ethical, time, and cost prohibitive; not for early-stage screening. |
Table 2: Correlation of Key Molecular Properties with BBB Permeability Across Platforms
| Molecular Property | In Silico Model Weighting | In Vitro Papp Correlation | In Situ Kin Correlation | Optimal Range for CNS Penetration |
|---|---|---|---|---|
| Molecular Weight (Da) | High (Negative) | Strong Negative (LogPapp ∝ -LogMW) | Strong Negative | Typically < 450 Da |
| LogP (Lipophilicity) | Very High (Optimum ~2-5) | Bell-shaped curve (Optimum ~2-4) | Positive, up to a plateau | 2.0 - 5.0 |
| Polar Surface Area (Ų) | High (Negative) | Strong Negative | Strong Negative | < 60-70 Ų |
| Hydrogen Bond Donors | High (Negative) | Strong Negative | Strong Negative | ≤ 3 |
| P-gp Substrate Probability | Moderate (Negative if efflux) | Directly measured via Efflux Ratio | Can be calculated (Kin,w/Kin) | Low probability desired |
Visualization of Workflows and Relationships
Title: BBB Permeability Prediction Decision Workflow
Title: Key Property Impact on BBB Permeability Pathways
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for BBB Permeability Research
| Item | Function/Brief Explanation | Example Vendor/Catalog |
|---|---|---|
| hCMEC/D3 Cell Line | Immortalized human cerebral microvascular endothelial cells; standard for in vitro BBB models. | Merck (SCC066) |
| iPSC-Derived BMEC Kits | Induced pluripotent stem cell-derived brain endothelial cells; more physiological phenotype. | STEMCELL Tech (Catalog #100-0523) |
| Collagen IV & Fibronectin | Extracellular matrix proteins for coating Transwell inserts to promote cell adhesion and barrier formation. | Corning (354233, 354008) |
| Transwell Permeable Supports | Polyester or polycarbonate inserts with porous membrane (0.4µm, 12-well) for culturing cell monolayers. | Corning (3460) |
| EVOM Voltohmmeter | Instrument for daily measurement of Transendothelial Electrical Resistance (TEER) to monitor barrier integrity. | World Precision Instruments |
| [14C]-Sucrose or Sodium Fluorescein | Paracellular integrity markers with low passive permeability; used to validate monolayer tightness. | American Radiolabeled Chemicals |
| Rhodamine 123 or Digoxin | Prototypical P-glycoprotein (P-gp) substrates; used to assess functional efflux transporter activity. | Sigma-Aldrich (R8004, D6003) |
| P-gp Inhibitor (e.g., Zosuquidar, Elacridar) | Specific inhibitor used in in vitro assays to confirm P-gp-mediated efflux of test compounds. | Tocris Bioscience (2314, 3300) |
| LC-MS/MS System | Gold-standard analytical platform for quantifying unlabeled test compound concentrations in permeability assays. | Sciex, Agilent, Waters |
| Radioactive Scintillation Counter | For quantifying radiolabeled tracers in in situ perfusion and some in vitro flux studies. | PerkinElmer |
This whitepaper examines the critical roles of molecular weight (MW) and lipophilicity (quantified by LogP) in the design and development of biologic and novel therapeutic modalities, with a specific focus on their implications for blood-brain barrier (BBB) permeability. While traditional small-molecule drug design relies heavily on Lipinski’s Rule of Five, biologics and novel modalities operate under distinct physicochemical paradigms. This guide provides a technical deep dive into how MW and LogP influence the pharmacokinetics, biodistribution, and ultimately, the therapeutic potential of antibodies, antisense oligonucleotides (ASOs), and other emerging modalities.
The historical framework for predicting drug-likeness and passive membrane permeability has been governed by parameters like MW (<500 Da) and LogP (between -0.4 and +5.6). However, the rise of biologics and novel modalities has necessitated a paradigm shift. These agents, with MW often exceeding 10 kDa and possessing highly polar/charged structures, cannot rely on passive diffusion. Their interaction with biological barriers, particularly the BBB, is dictated by specialized transport mechanisms and formulation strategies, making MW and LogP key, but differently interpreted, design factors.
Quantitative Landscape of Modalities
The table below summarizes the characteristic MW and LogP ranges for different therapeutic classes, highlighting their divergence from small molecules.
Table 1: Molecular Weight and LogP Ranges Across Therapeutic Modalities
| Therapeutic Modality | Typical MW Range | Characteristic LogP/Distribution Coefficient | Primary BBB Permeability Mechanism |
|---|---|---|---|
| Traditional Small Molecules | 200 - 500 Da | 1 - 3 (clogP) | Passive diffusion |
| Monoclonal Antibodies (mAbs) | ~150 kDa | Highly negative (cLogP < -5)* | Receptor-mediated transcytosis (RMT) |
| Antibody-Drug Conjugates (ADCs) | ~150 - 200 kDa | Variable (depends on linker/warhead) | Limited; RMT for antibody, warhead release |
| Antisense Oligonucleotides (ASOs) | 6 - 10 kDa | Highly negative (LogD ~ -5 to -1) | Very limited; some carrier-mediated/adsorptive transcytosis |
| siRNAs | ~13 kDa | Highly negative | Endocytosis/transcytosis (formulation-dependent) |
| Peptides | 1 - 10 kDa | Variable (can be engineered) | Paracellular leakage, RMT, or carrier-mediated |
| PROTACs | 700 - 1200 Da | Often low (cLogP ~1-4) | Passive diffusion (challenged by high MW) |
Calculated LogP is not meaningful for intact mAbs; surface hydrophilicity is high. *LogD at physiological pH is more relevant due to ionizable phosphate groups.
Modality-Specific Analysis
Monoclonal Antibodies (mAbs)
mAbs are large, hydrophilic proteins. Their high MW (~150 kDa) and negative effective LogP preclude passive diffusion. BBB penetration is minimal (<0.1% of injected dose typically reaches the brain). Strategies to overcome this involve engineering for Receptor-Mediated Transcytosis (RMT) via endogenous BBB receptors (e.g., Transferrin Receptor, Insulin Receptor).
Experimental Protocol: Assessing mAb Brain Uptake via RMT
- Objective: Quantify brain uptake of a candidate anti-Transferrin Receptor (TfR) bispecific antibody.
- Materials: Radiolabeled (e.g., Iodine-125) or fluorescently labeled test antibody, control IgG, wild-type mice.
- Method:
- Administration: Inject a known dose intravenously.
- Circulation: Allow a specific time for circulation (e.g., 24 hours).
- Perfusion: Euthanize and transcardially perfuse with saline to clear intravascular blood.
- Harvesting: Remove brain and homogenize.
- Quantification: Measure radioactivity or fluorescence in homogenate vs. plasma.
- Calculation: Express data as %Injected Dose per gram of brain tissue (%ID/g) or brain/plasma ratio.
- Key Analysis: Compare uptake of the TfR-targeting antibody to the control IgG. Significantly higher %ID/g indicates successful RMT engagement.
Antisense Oligonucleotides (ASOs)
ASOs are single-stranded DNA/RNA analogs with a phosphorothioate backbone, conferring negative charge and moderate lipophilicity (LogD ~ -1 to -5). Their MW (~6-10 kDa) and charge prevent passive BBB crossing. Naked ASOs have minimal CNS penetration. Chemical modifications (e.g., increasing ligand conjugation like GalNAc for liver, not brain) or formulations (e.g., lipid nanoparticles) are required for CNS delivery. Intrathecal injection is the current clinical route for CNS targets.
Experimental Protocol: Measuring ASO Brain Exposure after Intrathecal Delivery
- Objective: Determine concentration and distribution of an ASO in brain regions after intrathecal bolus injection.
- Materials: Fluorescently or affinity-tagged ASO, rodent model, micro-sampling probe or tissue harvest tools.
- Method:
- Dosing: Perform intrathecal injection (e.g., via cisterna magna puncture) of ASO.
- Time Course: Sacrifice animals at multiple time points (e.g., 1h, 24h, 7d).
- Tissue Collection: Dissect brain into regions (cortex, hippocampus, spinal cord).
- Sample Processing: Homogenize tissues and extract ASO.
- Quantification: Use hybridization-based ELISA (e.g., sandwich immunoassay with anti-ASO capture/detection) or LC-MS/MS.
- Data Presentation: Report ng of ASO per mg of tissue for each region over time.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for BBB Permeability Research on Novel Modalities
| Reagent / Material | Function/Description | Key Application |
|---|---|---|
| In Vitro BBB Models (e.g., hCMEC/D3 cells, iPSC-derived BMECs) | Cultured brain endothelial cells, often in Transwell systems, to model barrier integrity (TEER) and permeability. | Screening passive/active transport of modalities in a controlled, reductionist system. |
| TfR-Binding Antibodies (e.g., clone 8D3) | Antibodies targeting the murine transferrin receptor, used as positive controls or components for engineering. | Validating RMT pathways in rodent models. |
| Phosphorothioate-Modified Control ASO | A non-targeting ASO with a standard backbone, often fluorescently labeled (e.g., Cy3). | Tracing biodistribution and establishing baseline pharmacokinetics of oligonucleotides. |
| LC-MS/MS with Protein/Oligo Capture | Analytical platform coupled with specific capture (SPE, immunoaffinity) for large molecule bioanalysis. | Quantifying intact biologics and their metabolites in complex matrices like brain homogenate. |
| Brain Perfusion Buffer | Isotonic, oxygenated physiological buffer (e.g., Krebs-Henseleit). | Performing in situ brain perfusion studies to isolate brain uptake from systemic PK factors. |
| Species-Specific FcRn Proteins | Recombinant FcRn for surface plasmon resonance (SPR) or affinity chromatography. | Measuring binding affinity critical for antibody half-life, which impacts brain exposure time. |
| Radioisotope or Fluorescent Labels (I-125, Alexa Fluor 680, near-IR dyes) | Tags for sensitive in vivo imaging and ex vivo tissue quantification. | Tracking real-time or terminal biodistribution of high-MW therapeutics. |
Integrated Analysis for Novel Modality Design
The interplay of MW and LogP must be contextualized within the delivery mechanism. For biologics, MW dictates the rate of convection and interstitial diffusion within tissues, while surface hydrophilicity/charge (related to LogP) influences solubility, stability, and off-target binding. For ASOs, backbone modifications alter LogD and protein binding, which affects plasma half-life and tissue distribution. The overarching goal for CNS targets is to optimize the "Brain Availability" metric, which is a function of plasma exposure, BBB permeation (via active transport), and brain parenchymal penetration.
MW and LogP remain foundational descriptors, but their interpretation is modality-dependent. For biologics and novel modalities, these parameters are not simple filters but levers to be engineered in concert with active transport biology. Future research is directed towards: 1) Predictive in silico models for active transport rates, 2) Advanced chemical platforms to tune LogD and charge distribution of oligonucleotides, and 3) Novel protein engineering to create shuttle systems with optimal MW and binding affinity for RMT. Success in CNS drug development will hinge on integrating this refined understanding of MW and LogP into the rational design of next-generation therapeutics.
Within the ongoing thesis on Blood-Brain Barrier (BBB) permeability factors, the relationship between molecular weight (MW), lipophilicity (commonly represented by LogP), and passive diffusion remains a cornerstone. While the "Lipinski's Rule of 5" established foundational guidelines, subsequent research has revealed significant complexities and common misinterpretations. This guide details the pitfalls in oversimplifying these parameters, leading to costly failures in central nervous system (CNS) drug development.
Core Principles and Common Misinterpretations
The simplistic view posits that lower MW and higher LogP guarantee superior BBB permeability. Failures arise from ignoring confounding factors.
Pitfall 1: Over-reliance on Calculated LogP (clogP) Calculated LogP values often diverge significantly from experimentally measured values (mLogP), especially for complex molecules with intramolecular hydrogen bonding or charged groups.
Pitfall 2: Ignoring Molecular Descriptors Beyond MW and LogP Polar Surface Area (PSA), hydrogen bond donor/acceptor count, and flexibility (number of rotatable bonds) are critical determinants often overshadowed by MW/LogP.
Pitfall 3: Assuming Passive Diffusion is the Sole Mechanism Focusing solely on passive diffusion ignores the role of active influx transporters (e.g., LAT1, GLUT1) and efflux pumps (notably P-glycoprotein, P-gp). A molecule with "ideal" MW and LogP may be a P-gp substrate and thus effectively excluded from the brain.
Pitfall 4: Extrapolating from Octanol-Water to Biological Membranes The octanol-water partition system (LogP) is a simplistic model. It fails to account for specific interactions with membrane phospholipids, cholesterol, and the asymmetric nature of the BBB endothelium.
Table 1: Comparison of Key Physicochemical Properties for CNS vs. Non-CNS Drugs (Representative Analysis)
| Property | CNS Drugs (Median) | Peripherally Acting Drugs (Median) | Common Misinterpretation Threshold |
|---|---|---|---|
| Molecular Weight (Da) | 305.3 | 349.6 | Arbitrary <500 Da cutoff |
| Calculated LogP (clogP) | 2.8 | 2.5 | "Higher is always better" |
| Topological PSA (Ų) | 44.8 | 75.6 | Often overlooked |
| Hydrogen Bond Donors | 1.0 | 1.6 | Overlooked in MW/LogP focus |
| Rotatable Bonds | 4.0 | 6.0 | Rarely considered |
Data synthesized from recent literature reviews and analyses of marketed drug databases (2020-2023).
Table 2: Impact of P-gp Efflux on Brain Penetration Despite Favorable MW/LogP
| Compound | MW (Da) | mLogP | P-gp Substrate? | Brain/Plasma Ratio (in vivo, rat) | Outcome |
|---|---|---|---|---|---|
| Loperamide | 477 | 5.3 | Yes | <0.1 | Failure: Efflux dominates |
| Caffeine | 194 | -0.1 | No | ~1.0 | Success: Despite low LogP |
| Verapamil | 455 | 3.8 | Yes (also inhibitor) | Variable (0.2-2) | Complex: Dose-dependent |
Experimental Protocols
Protocol: Determination of Experimental LogD (pH 7.4)
Objective: To measure the distribution coefficient (LogD) at physiological pH, providing a more relevant lipophilicity metric than LogP.
- Buffer Preparation: Prepare 0.15 M phosphate buffer, pH 7.4.
- Partitioning System: Combine 3 mL of octanol (pre-saturated with buffer) and 3 mL of buffer (pre-saturated with octanol) in a glass vial.
- Spiking: Add a known mass of test compound to achieve a concentration well below saturation.
- Equilibration: Vortex mix for 10 minutes, then centrifuge at 3000 rpm for 15 minutes for complete phase separation.
- Quantification: Carefully separate phases. Analyze the concentration of the compound in each phase using a validated analytical method (e.g., HPLC-UV/LC-MS).
- Calculation: LogD = Log₁₀ (Concentration in octanol / Concentration in buffer).
Protocol: Parallel Artificial Membrane Permeability Assay (PAMPA-BBB)
Objective: To assess passive transcellular permeability specifically predictive of BBB penetration.
- Membrane Preparation: Dissect 2% (w/v) porcine brain lipid (PBL) in dodecane. Filter (0.45 µm).
- Plate Assembly: Inject 5 µL of the lipid solution onto a 0.45 µm hydrophobic PVDF filter of the donor plate. Add 300 µL of test compound (100 µM in pH 7.4 buffer) to the donor well.
- Acceptor Plate: Fill the acceptor plate wells with 300 µL of pH 7.4 buffer (with 5% DMSO to maintain sink conditions).
- Incubation: Assemble the sandwich plate and incubate at 25°C for 4 hours.
- Quantification: Analyze compound concentration in donor and acceptor wells post-incubation via HPLC-UV/LC-MS.
- Calculation: Determine permeability, Pₑ (x 10⁻⁶ cm/s). Validate with reference compounds (e.g., Verapamil: High Pₑ; Sucrose: Low Pₑ).
Protocol: Cell-Based Assay for P-gp Efflux (MDCK-MDR1)
Objective: To determine if a compound is a substrate for the P-glycoprotein efflux transporter.
- Cell Culture: Culture MDCKII cells stably transfected with human MDR1 gene. Seed cells on 24-well Transwell inserts at high density. Culture for 5-7 days until transepithelial electrical resistance (TEER) > 1500 Ω·cm².
- Bidirectional Transport: Prepare compound (10 µM) in transport buffer (HBSS, 10 mM HEPES, pH 7.4).
- A-to-B (Apical to Basolateral): Add compound to apical chamber, buffer to basolateral.
- B-to-A (Basolateral to Apical): Add compound to basolateral chamber, buffer to apical.
- With/Without Inhibitor: Include a control set with a P-gp inhibitor (e.g., 10 µM GF120918 or 100 µM verapamil) in both chambers.
- Incubation & Sampling: Incubate at 37°C. Sample from receiver chamber at 60, 120 minutes. Replace with fresh buffer.
- Analysis & Calculation: Quantify samples by LC-MS/MS. Calculate apparent permeability (Papp) for each direction. Compute Efflux Ratio = Papp(B-to-A) / Papp(A-to-B). An ER > 2 that is reduced >50% by inhibitor indicates P-gp substrate.
Diagrams
Diagram 1: Factors Influencing BBB Permeability
Diagram 2: Integrated Permeability & Efflux Workflow
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagent Solutions for MW/LogP and Permeability Research
| Item | Function / Application | Key Consideration |
|---|---|---|
| Phosphate Buffered Saline (PBS), pH 7.4 | Universal aqueous phase for LogD/PAMPA; cell culture wash. | Must be isotonic and at physiological pH for relevant data. |
| n-Octanol (Buffer-Saturated) | Organic phase for experimental LogP/LogD determination. | Pre-saturation with buffer is critical to prevent water uptake and volume shifts. |
| Porcine Brain Lipid (PBL) Extract | Lipid mixture for PAMPA-BBB to mimic BBB endothelial membrane. | Use high-purity, defined extracts for assay reproducibility. |
| MDCKII-MDR1 Cell Line | Mammalian cell line for assessing P-gp mediated efflux. | Regularly check MDR1 expression (e.g., by qPCR) and monolayer integrity (TEER). |
| P-glycoprotein Inhibitors (e.g., GF120918, Verapamil) | Pharmacological tools to confirm P-gp substrate status in transport assays. | Use at non-cytotoxic, selective concentrations. Include solvent controls. |
| LC-MS/MS Grade Solvents (MeOH, ACN, Water) | For sample preparation and compound quantification in permeability/transport assays. | High purity minimizes background interference and ion suppression. |
| Reference Compounds (e.g., Caffeine, Verapamil, Sucrose) | High/low permeability and P-gp substrate controls for assay validation. | Run in every assay batch to ensure system suitability and inter-experiment comparability. |
The efficacy of central nervous system (CNS)-targeted therapeutics is fundamentally constrained by the blood-brain barrier (BBB). This article details emerging computational and experimental frameworks designed to quantitatively integrate key permeability factors—specifically molecular weight (MW) and lipophilicity, often expressed as log P (or log D at physiological pH)—into unified Pharmacokinetic/Pharmacodynamic (PK/PD) models. This integration is critical for rational CNS drug design, enabling the prediction of not just brain exposure but also the resultant pharmacological effect over time.
Core Permeability Factors: Molecular Weight and Lipophilicity
The passive diffusion of compounds across the BBB is predominantly governed by physicochemical properties. Empirical rules, such as Lipinski's Rule of 5, are adapted for CNS penetration, often emphasizing lower MW and moderate lipophilicity. Excessive lipophilicity can impair solubility and increase non-specific binding, reducing free brain concentration.
Table 1: Impact of Physicochemical Properties on BBB Permeability and PK/PD
| Property | Optimal Range for BBB Penetration | Impact on PK Parameters | Influence on PD Modeling |
|---|---|---|---|
| Molecular Weight (Da) | Typically <450-500 | Affects volume of distribution (Vd), clearance (CL). | Determines rate of target site access; influences effect compartment equilibration rate (k~e0~). |
| Lipophilicity (Log D~7.4~) | 1-3 | Increases plasma protein binding, tissue penetration, and metabolic clearance. | Critical for estimating unbound brain concentration (C~u,brain~), the driver of pharmacodynamic effect. |
| Passive Permeability (P~app~ in cm/s) | >5 x 10^-6^ (Caco-2/MDCK) | Informs absorption rate constant (K~a~) and distribution. | Directly linked to the transfer rate constant between central and brain effect compartments (K~in~, K~out~). |
Integrative PK/PD Modeling Frameworks
The integration moves beyond simple permeability metrics to dynamic, mechanistic models.
Core Model Structures
- Direct Link PK/PD Models: Applied when plasma concentration is in rapid equilibrium with the brain effect site. Suitable for compounds with high passive permeability.
- Indirect Link PK/PD Models: Incorporate a hypothetical effect compartment to account for a temporal dissociation between plasma PK and brain PD. The permeability rate constants (K~in~, K~out~) can be parameterized using in vitro permeability data.
- Physiologically-Based Pharmacokinetic (PBPK) Brain Models: These models incorporate physiological compartments (brain vascular, endothelial, interstitial) and explicitly model transcellular passive diffusion (a function of MW/log P) and active transport processes.
Key Experimental Protocols for Model Parameterization
Protocol A: In Vitro BBB Permeability Assay for PK/PD Input
- Objective: Determine the apparent permeability (P~app~) of a compound to derive distribution rate constants for PK/PD models.
- Methodology:
- Utilize a monolayer of brain endothelial cells (e.g., hCMEC/D3 cell line) or MDCK cells on a transwell insert.
- Add test compound to the donor compartment (apical for blood-to-brain assay).
- Sample from the acceptor compartment at timed intervals (e.g., 15, 30, 60, 90, 120 min).
- Quantify compound concentration using LC-MS/MS.
- Calculate P~app~:
P_app = (dQ/dt) / (A * C_0), where dQ/dt is the steady-state flux, A is the filter area, and C~0~ is the initial donor concentration. - Correlate P~app~ with log D and MW using established algorithms (e.g., AlogPS) to predict in vivo K~in~.
Protocol B: In Vivo Microdialysis for Free Brain Concentration
- Objective: Measure time-course of unbound brain concentration (C~u,brain~) for direct PK/PD linking.
- Methodology:
- Surgically implant a microdialysis probe into the brain region of interest (e.g., striatum, hippocampus) in a rodent model.
- Perfuse the probe with artificial cerebrospinal fluid (aCSF) at a low flow rate (e.g., 0.5-2 µL/min).
- Administer the test compound systemically (IV, PO).
- Collect dialysate fractions at frequent intervals post-dose.
- Analyze dialysate (C~dialysate~) and concurrent plasma for compound concentration.
- Calculate C~u,brain~:
C_u,brain = C_dialysate / Recovery. In vivo recovery is determined via retrodialysis or no-net-flux methods. - Use C~u,brain~ vs. time profile as the direct PK driver for an Emax or other PD model.
Visualization of Key Frameworks
Title: Integrating Permeability Factors into PK/PD Modeling Frameworks
Title: Workflow for Building a Permeability-Informed PK/PD Model
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Research Reagents & Materials
| Item | Function in Permeability/PK/PD Research | Example/Supplier (Illustrative) |
|---|---|---|
| hCMEC/D3 Cell Line | Immortalized human cerebral microvascular endothelial cells; gold standard for in vitro BBB permeability studies. | Merck Millipore (SCC066) |
| Transwell Permeable Supports | Polyester or polycarbonate membrane inserts for growing cell monolayers and performing permeability assays. | Corning (e.g., 3460) |
| LC-MS/MS System | Essential for sensitive and specific quantification of drug concentrations in biological matrices (plasma, dialysate, homogenate). | Sciex, Waters, Agilent |
| Brain Microdialysis Kits | Sterile probes and cannulas for in vivo sampling of unbound brain interstitial fluid in rodents. | Harvard Apparatus, CMA Microdialysis |
| Artificial Cerebrospinal Fluid (aCSF) | Isotonic perfusion fluid for microdialysis experiments, mimicking the ionic composition of brain extracellular fluid. | Tooris Bioscience (3525) |
| PBPK/PD Modeling Software | Platforms for building, simulating, and fitting integrative mechanistic models (e.g., permeability-limited brain compartments). | GastroPlus, Simcyp Simulator, Berkeley Madonna |
| Log P/D Prediction Software | Computational tools for estimating lipophilicity from chemical structure, used in early design stages. | ACD/Percepta, ChemAxon, MoKa |
Conclusion
Molecular weight and lipophilicity remain the cornerstone physicochemical properties for predicting and optimizing BBB permeability, underpinning rational CNS drug design. Foundational rules provide essential heuristics, but modern drug discovery requires their integration with sophisticated methodological tools, proactive troubleshooting strategies, and rigorous in vivo validation. The future lies in advanced multi-parameter optimization models that balance permeability with other critical ADMET properties, and in expanding these principles to novel therapeutic modalities. A nuanced, data-driven understanding of MW and LogP, beyond simplistic rules, is imperative for translating neurotherapeutic candidates from bench to bedside successfully.