PBPK Modeling for Monoclonal Antibodies and Therapeutic Proteins: A Comprehensive Guide for Drug Development
This article provides a detailed exploration of Physiologically Based Pharmacokinetic (PBPK) modeling for large molecule therapeutics, including monoclonal antibodies and other proteins.
PBPK Modeling for Monoclonal Antibodies and Therapeutic Proteins: A Comprehensive Guide for Drug Development
Abstract
This article provides a detailed exploration of Physiologically Based Pharmacokinetic (PBPK) modeling for large molecule therapeutics, including monoclonal antibodies and other proteins. It establishes the foundational principles that distinguish large molecule PBPK from traditional small molecule approaches, outlines current methodologies and real-world applications in drug development, addresses common challenges and optimization strategies, and examines validation frameworks and comparative analyses with emerging techniques. Tailored for researchers, scientists, and drug development professionals, this guide synthesizes current industry standards and recent advances to offer actionable insights for integrating PBPK into the biologics pipeline.
Beyond Small Molecules: Laying the Groundwork for Large Molecule PBPK Modeling
Defining the Unique PBPK Landscape for mAbs and Therapeutic Proteins
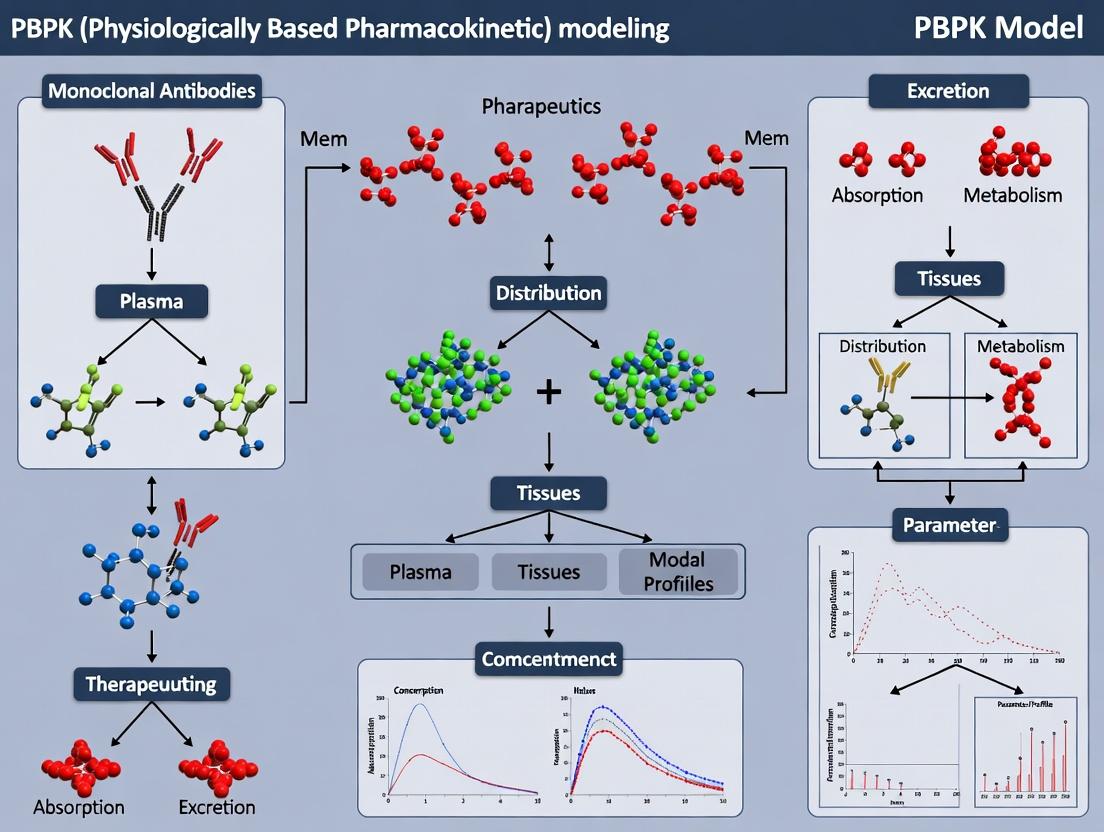
Physiologically-based pharmacokinetic (PBPK) modeling for monoclonal antibodies (mAbs) and therapeutic proteins (TPs) requires specialized frameworks distinct from small molecules. Their unique disposition is governed by large size, target-mediated drug disposition (TMDD), Fc-mediated recycling, and lymphatic transport. This note outlines the core components, data requirements, and applications of such models within a broader thesis on advancing biologic drug development.
Key Applications:
- First-in-Human (FIH) Dose Prediction: Scaling from preclinical species using allometric principles incorporating FcRn affinity and target abundance.
- Predicting TMDD: Understanding non-linear PK when drug-target binding is a major clearance pathway.
- Assessing Drug-Drug Interactions (DDIs): For cytokines or mAbs that modulate target or FcRn expression.
- Pediatric and Special Population Scaling: Incorporating age-dependent changes in physiology (e.g., lymph flow, FcRn expression).
- Bio-distribution to Target Tissues: Predicting exposure at sites of action (e.g., tumors, synovial fluid).
Core Quantitative Data for Model Parameterization
Table 1: Key Physiological Parameters for mAb PBPK Models
| Parameter | Typical Value (Human) | Source/Comment |
|---|---|---|
| Plasma Volume | ~3 L | Standard human physiology |
| Lymph Flow Rate | 0.2 - 0.5 L/h | Critical for interstitial distribution |
| Vascular Reflection Coefficient (σv) | 0.82 - 0.94 | Varies by tissue; governs convection |
| Lymphatic Reflection Coefficient (σL) | 0.2 - 0.3 | Assumed lower than σv |
| Endosomal pH | 6.0 - 6.5 | Critical for FcRn binding/recycling |
| Plasma FcRn Concentration | 0.4 - 0.6 µM | High-affinity binding site |
| IgG Plasma Half-Life | ~21 days | Baseline for FcRn salvage efficiency |
Table 2: Common Drug-Specific Parameters for mAbs/TPs
| Parameter | Typical Range | Method of Estimation |
|---|---|---|
| Target Affinity (KD) | pM - nM | Surface Plasmon Resonance (SPR) |
| Target Expression (Rtot) | pmol/g tissue | Quantitative biodistribution, PCR |
| Internalization Rate (kint) | 0.1 - 1.0 h⁻¹ | Cell-based assays with labeled drug |
| FcRn Affinity at pH 6.0 (KD) | 100 - 500 nM | SPR at endosomal pH |
| Non-specific Linear Clearance | 0.01 - 0.05 L/h | FcRn knockout animal studies |
Detailed Experimental Protocols for Critical Data Generation
Protocol 1: Quantifying Target Abundance (Rtot) via Radiolabeled Ligand Binding Assay
Objective: To determine the total target expression concentration in homogenates of relevant tissues.
Materials: Cryopreserved human tissue homogenates, [¹²⁵I]-labeled therapeutic mAb/TP, unlabeled competitor (same mAb), binding buffer, gamma counter, filtration manifold.
Procedure:
- Prepare a dilution series of tissue homogenate in binding buffer.
- Incubate homogenates with a fixed, trace concentration of [¹²⁵I]-mAb in the presence (non-specific binding) or absence (total binding) of excess unlabeled mAb.
- Incubate for 4-16 hours at 4°C to reach equilibrium.
- Separate bound from free radioligand using rapid vacuum filtration over GF/B filters.
- Wash filters 3x with ice-cold buffer.
- Quantify filter-bound radioactivity using a gamma counter.
- Data Analysis: Calculate specific binding (Total - Non-specific). Fit data to a saturable binding model to derive Bmax (Rtot) and KD.
Protocol 2: Determining FcRn Affinity (KD) via Surface Plasmon Resonance (SPR)
Objective: To measure the pH-dependent binding affinity of the mAb to human FcRn.
Materials: Biacore or equivalent SPR instrument, CMS sensor chip, recombinant human FcRn, anti-FcRn antibody for capture, running buffers (pH 7.4 and pH 6.0), serial dilutions of mAb analyte.
Procedure:
- Immobilize an anti-FcRn antibody on the CMS chip via standard amine coupling.
- Capture a consistent level of recombinant FcRn onto the antibody surface at pH 7.4.
- Switch to pH 6.0 running buffer to mimic endosomal conditions.
- Inject a series of mAb concentrations (e.g., 10 nM to 1 µM) over the FcRn surface at pH 6.0.
- Monitor association, then dissociate in pH 6.0 buffer.
- Regenerate the surface with a pH 7.4 buffer pulse to release FcRn/mAb complex.
- Data Analysis: Fit the resulting sensograms globally to a 1:1 Langmuir binding model using the instrument software to derive the association (kon) and dissociation (koff) rates at pH 6.0. Calculate KD = koff/kon.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for mAb PBPK Research
| Item | Function |
|---|---|
| Recombinant Human FcRn | Critical for in vitro assessment of mAb recycling and half-life extension potential. |
| Biacore Series S Sensor Chip CMS | Gold-standard for label-free, real-time kinetics (SPR) of protein-protein interactions. |
| Cryopreserved Human Tissue Homogenates | Provide physiologically relevant matrices for target expression and binding studies. |
| [¹²⁵I] Sodium Iodide | Radiolabel for sensitive quantitative biodistribution and ex vivo binding studies. |
| pH-Sensitive Cell Lines (e.g., engineered HEK293) | Enable cell-based internalization and FcRn recycling assays under controlled pH conditions. |
| PBPK Software (e.g., GastroPlus, Simbiology) | Platforms with dedicated mAb/TP modules for model construction and simulation. |
Visualizing Key Pathways and Workflows
Diagram 1: mAb PBPK Disposition Pathways
Diagram 2: Workflow for Model Development & Validation
This application note details key biological processes relevant to the PBPK modeling of monoclonal antibodies (mAbs) and therapeutic proteins. Within the context of a predictive PBPK framework, understanding FcRn-mediated recycling and Target-Mediated Drug Disposition (TMDD) is critical for accurately simulating the complex, nonlinear pharmacokinetics of these biologics. This document provides experimental protocols and quantitative data summaries to support the characterization of these processes in drug development.
FcRn-Mediated Recycling: Mechanisms & Experimental Characterization
The neonatal Fc receptor (FcRn) is a central regulator of IgG and albumin homeostasis. It protects these proteins from lysosomal degradation by binding them in acidic endosomes (pH ~6.0) and recycling them back to the cell surface for release at neutral pH (~7.4). This process significantly extends the serum half-life of mAbs.
Table 1: Key Parameters for FcRn-Mediated Recycling of mAbs
| Parameter | Typical Value Range | Impact on PK |
|---|---|---|
| FcRn-IgG Binding Affinity (Kd at pH 6.0) | 100 - 600 nM | Higher affinity increases half-life, but very high affinity can saturate system. |
| Plasma Half-life (Human IgG1) | ~21 days | Directly influenced by recycling efficiency. |
| Endosomal pH for Binding | 5.5 - 6.5 | Critical for pH-dependent binding/release cycle. |
| Serum IgG Concentration | ~10 mg/mL | Endogenous IgG competes with therapeutic mAb for FcRn binding. |
Protocol 1:In VitroFcRn Binding Affinity Assay (SPR/BLI)
Objective: Determine the pH-dependent binding kinetics of a mAb to human FcRn. Materials:
- Biacore T200/Blitz System: For Surface Plasmon Resonance (SPR) or Bio-Layer Interferometry (BLI).
- Recombinant Human FcRn Protein: Purified, biotinylated for sensor immobilization.
- mAb Analyte: Test mAb at high purity.
- HBS-EP Buffer (pH 7.4): Running buffer for dissociation phase.
- MES Buffer (pH 5.5 - 6.0): Running buffer for association phase.
- Regeneration Solution: 0.1 M Glycine, pH 2.0 - 2.5.
Procedure:
- Immobilization: Capture biotinylated FcRn onto a streptavidin (SA) sensor chip (SPR) or dip (BLI).
- pH-Specific Binding Cycle: a. Equilibrate system with MES buffer (pH 5.8). b. Inject a dilution series of the mAb (e.g., 12.5 - 400 nM) over the FcRn surface for association. c. Switch to HBS-EP buffer (pH 7.4) to initiate and monitor dissociation.
- Regeneration: Inject glycine pH 2.0 for 30 seconds to remove bound mAb.
- Data Analysis: Fit sensorgrams globally using a 1:1 Langmuir binding model to derive association (ka) and dissociation (kd) rate constants, and calculate KD (kd/ka).
Diagram: FcRn Recycling Pathway
Diagram Title: FcRn-Mediated IgG Recycling and Salvage Pathway
The Scientist's Toolkit: FcRn Research
Table 2: Essential Reagents for FcRn Studies
| Reagent | Function & Explanation |
|---|---|
| Recombinant Human FcRn (Biotinylated) | Enables consistent immobilization on SPR/BLI sensors for controlled binding assays. |
| pH-Specific Assay Buffers (MES, HEPES) | Mimics the pH gradient of the endosomal cycle (pH 6.0 for binding, pH 7.4 for release). |
| Human Endothelial Cell Lines (e.g., HUVEC) | Express endogenous FcRn; used for transcytosis and cellular recycling studies. |
| FcRn Knockout Mouse Model | In vivo model to definitively assess the role of FcRn in mAb pharmacokinetics. |
| Anti-FcRn Blocking Antibodies | Tools to inhibit FcRn function in vitro and in vivo to study impact on mAb half-life. |
Target-Mediated Drug Disposition (TMDD): Principles & Characterization
TMDD describes nonlinear PK observed when a significant portion of a therapeutic biologic is bound to a high-affinity, pharmacologically relevant target with limited capacity. This leads to dose- and time-dependent PK, characterized by rapid initial clearance at low doses that saturates at higher doses. It involves binding, internalization, and degradation of the drug-target complex.
Table 3: Key Parameters in TMDD Models
| Parameter | Symbol | Typical Units | Description |
|---|---|---|---|
| Target Concentration | Rtot | nmol/L | Total target density (membrane + soluble). |
| Drug-Target Binding Affinity | KD | nM | Equilibrium dissociation constant. |
| Internalization Rate Constant | kint | h-1 | Rate of drug-target complex elimination. |
| Target Synthesis Rate | ksyn | nmol/L/h | Zero-order rate of new target production. |
| Target Degradation Rate | kdeg | h-1 | First-order rate of natural target turnover. |
Protocol 2: Characterizing Cell-Based TMDD Parameters
Objective: Determine target binding affinity (KD) and internalization rate (kint) using a target-expressing cell line. Materials:
- Target-Expressing Cells: Stably transfected cell line.
- Radio- or Fluoro-labeled Therapeutic Protein: e.g., [¹²⁵I]-mAb or Alexa Fluor 647-mAb.
- Binding Buffer: Ice-cold PBS with 1% BSA.
- Acid Wash Buffer: 0.2 M acetic acid, 0.5 M NaCl (pH ~2.5).
- Lysis Buffer: 1% Triton X-100 in PBS.
- Gamma Counter or Flow Cytometer.
Procedure: Part A: Saturation Binding for KD and Rtot
- Plate cells in 24-well plates.
- At confluence, incubate with increasing concentrations of labeled drug in binding buffer at 4°C for 4-6 hours (prevents internalization).
- Wash cells 3x with ice-cold buffer.
- Lyse cells and measure cell-associated radioactivity/fluorescence.
- Perform nonlinear regression of specific binding vs. concentration to derive Bmax (Rtot) and KD.
Part B: Internalization Rate (kint)
- Incubate cells with a saturating concentration of labeled drug at 37°C for various time points (e.g., 0, 15, 30, 60, 120 min).
- At each time point, stop reaction on ice.
- Strip surface-bound drug with two 5-minute washes of acid wash buffer.
- Lyse cells and measure acid-resistant (internalized) radioactivity/fluorescence.
- Fit the time course of internalized drug to a first-order equation to estimate kint.
Diagram: TMDD Mechanism and PK Impact
Diagram Title: TMDD: Cellular Mechanism and Nonlinear PK
The Scientist's Toolkit: TMDD Research
Table 4: Essential Tools for TMDD Analysis
| Reagent/Tool | Function & Explanation |
|---|---|
| Target-Expressing Cell Lines | Provide a controlled system to measure binding and internalization kinetics in vitro. |
| Labeled Drug Conjugates (¹²⁵I, Alexa Fluor) | Enable quantitative tracking of drug distribution, binding, and uptake. |
| Soluble Target Protein (sAntigen) | Used in competition assays and to quantify free drug levels in PK studies. |
| Pharmacokinetic Software (e.g., NONMEM, Phoenix) | Essential for fitting complex TMDD models to in vivo concentration-time data. |
| Quantitative Whole-Body Autoradiography (QWBA) | Imaging technique to visualize tissue distribution and target engagement in vivo. |
Integration into PBPK Modeling
For whole-body PBPK models, parameters derived from these protocols (FcRn KD, kint, Rtot, ksyn, kdeg) are incorporated into tissue compartments. FcRn recycling is often modeled in endothelial cells of representative tissues, while TMDD is implemented in target-expressing tissues. This mechanistic integration allows for the prediction of human PK, inter-individual variability, and the design of optimal first-in-human dosing regimens.
Essential Structural Components of a Large Molecule PBPK Model
Within the broader thesis on advancing PBPK modeling for monoclonal antibodies (mAbs) and therapeutic proteins, defining the essential structural components of a large molecule PBPK model is critical. These models are distinct from small-molecule PBPK due to the complex physiology governing the disposition of biologics. This document outlines the core structural elements, provides protocols for their development, and details the necessary research toolkit.
Core Structural Components & System Parameters
Large molecule PBPK models are built upon physiological compartments representing key organs/tissues connected by vascular and lymphatic flows. The core components can be categorized into system-specific, drug-specific, and interaction-specific parameters.
Table 1: Essential Structural Components of a Large Molecule PBPK Model
| Component Category | Specific Parameter | Description & Typical Value/Source | Rationale in mAb/PBPK | |
|---|---|---|---|---|
| Physiological System | Organ Plasma Volumes | Blood/plasma volume of liver, spleen, muscle, etc. (e.g., Liver plasma vol: ~0.7 L) | From human physiology textbooks & population studies. | Defines the central volume of distribution and initial dilution space. |
| Vascular & Lymphatic Flow Rates | Blood flow (Q) between organs; lymph flow (L) from tissue interstitium. (e.g., Lymph flow from muscle: ~0.0002 L/h) | Literature values for human physiology. | Governs convective transport of mAbs between compartments. Key for describing lymphatic recirculation. | |
| Vascular Reflection Coefficients (σv) | Coefficient (0-1) for permeability of capillaries to large molecules. Muscle: ~0.95; Liver: ~0.1. | Estimated from experimental data or prior models. | Controls paracellular extravasation via pore theory. Tissue-specific. | |
| Lymphatic Reflection Coefficients (σL) | Similar to σv but for lymphatic capillaries. Often set equal to σv. | Assumed or fitted. | Impacts protein return from interstitium to plasma. | |
| Tissue Architecture | Endosomal Volume Fraction | Fraction of tissue volume occupied by endosomes. (e.g., ~0.01 of tissue volume) | From cell biology data, often a sensitive fitted parameter. | Critical for modeling intracellular catabolism via the FcRn salvage pathway. |
| Interstitial Volume Fraction | Fraction of tissue volume that is interstitial fluid. (e.g., Muscle: ~0.12; Skin: ~0.35) | Physiological literature. | Primary distribution space for mAbs outside the vasculature. | |
| Drug Properties | Molecular Weight | mAbs: ~150 kDa; Fusion proteins: variable. | Experimental data (e.g., SEC-MALS). | Impacts diffusion and renal filtration threshold. |
| Isoelectric Point (pI) | Net charge at physiological pH. (e.g., typical mAb pI: 7-9) | Calculated or measured (e.g., imaged cIEF). | Influences electrostatic interaction with charged endothelial glycocalyx and tissues (non-specific binding). | |
| Affinity to FcRn (KD) | Dissociation constant for Fc-FcRn binding at endosomal pH (6.0). (e.g., 0.5 - 2 µM) | Measured via surface plasmon resonance (SPR). | Determines efficiency of cellular recycling and half-life extension. | |
| Target Affinity (KD) | Binding to pharmacological target (e.g., soluble antigen, membrane receptor). | SPR or bio-layer interferometry. | Drives target-mediated drug disposition (TMDD), a key nonlinearity. | |
| Interaction & Turnover | FcRn Expression Level | Tissue concentration of FcRn (e.g., high in endothelium, muscle). | Quantitative proteomics or literature. | Scales the capacity of the salvage pathway. |
| Target Expression (Rtotal) | Target antigen density (molecules/cell) or soluble concentration. | Biomarker assays, qPCR, flow cytometry. | Essential for constructing TMDD component of the model. | |
| Endocytic Rate (kint) | Rate of nonspecific pinocytosis/internalization. (e.g., 0.1 - 0.3 day-1) | Fitted or derived from in vitro assays. | Drives cellular uptake for catabolism or recycling. | |
| Linear Clearance Rate | Non-saturable, non-target elimination (e.g., via catabolism in cells). | Fitted from in vivo PK data at supra-saturating doses. | Represents baseline elimination. |
Experimental Protocols for Parameter Estimation
Protocol 2.1: Determining FcRn Binding Affinity (KD) via Surface Plasmon Resonance (SPR)
Objective: To measure the pH-dependent binding affinity of a therapeutic mAb to human FcRn. Materials: See "Scientist's Toolkit" below. Workflow:
- Chip Preparation: Immobilize recombinant human FcRn protein on a CM5 sensor chip using standard amine coupling to achieve a density of ~5000 Response Units (RU).
- Running Buffer: Prepare HBS-EP+ buffer at two pH levels: pH 6.0 (endosomal) and pH 7.4 (physiological/plasma).
- Sample Preparation: Serially dilute the mAb analyte in pH 6.0 running buffer (e.g., 0.78 nM to 200 nM).
- Binding Cycle: At 25°C, inject mAb dilutions over the FcRn and reference surfaces for 180 seconds at a flow rate of 30 µL/min using pH 6.0 buffer.
- Dissociation: Switch to pH 7.4 buffer for 300 seconds to monitor dissociation.
- Regeneration: Inject a pulse of pH 7.4 buffer with 0.3 M NaCl for 30 seconds to fully regenerate the surface.
- Data Analysis: Subtract reference cell data. Fit the resulting sensorgrams to a 1:1 Langmuir binding model using the Biacore Evaluation Software to obtain the association rate (ka), dissociation rate (kd), and KD (kd/ka).
Protocol 2.2: Quantifying In Vivo Linear Clearance via High-Dose PK Study
Objective: To estimate the non-saturable, linear clearance (CL) of a mAb for PBPK model initialization. Workflow:
- Animal Dosing: Administer a single intravenous dose of the mAb to mice (or relevant species) at a level known to saturate all target-mediated pathways (e.g., 100 mg/kg). Use n=5-6 animals per time point.
- Serial Blood Sampling: Collect plasma samples at predefined time points (e.g., 0.083, 1, 6, 24, 72, 168, 336 hours post-dose).
- Bioanalysis: Quantify mAb concentrations in plasma using a validated ligand-binding assay (e.g., ELISA).
- Non-Compartmental Analysis (NCA): Calculate the terminal half-life (t1/2) and area under the curve (AUC0-inf) from the mean concentration-time profile.
- Clearance Calculation: Compute linear clearance: CL = Dose / AUC0-inf. This value represents the aggregate linear elimination parameter to be refined in the PBPK model.
Visualization of Model Structure and Key Pathways
Diagram 1: Full PBPK Model Structure for a mAb
Diagram 2: FcRn Salvage Pathway Mechanism
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Large Molecule PBPK Model Development
| Item | Function & Application | Example Vendor/Product |
|---|---|---|
| Recombinant Human FcRn Protein | Critical reagent for SPR assays to measure binding affinity (KD) at endosomal pH. | Sino Biological, Themo Fisher Scientific. |
| SPR Instrument & Chips | Platform for real-time, label-free analysis of biomolecular interactions (e.g., FcRn/mAb binding). | Cytiva (Biacore), Sartorius (Octet). |
| Human Tissue Biomarker Quantification Kits | To quantify target antigen expression levels (soluble or membrane-bound) in tissues/plasma. | R&D Systems ELISA kits, MSD assays. |
| Anti-idiotypic Antibodies | Essential capture/detection reagents for developing PK assays specific to the therapeutic mAb. | Custom generation from companies like Bio-Rad. |
| Physiologically-based PK/PD Modeling Software | Platform to code, simulate, and fit the PBPK model structure. | GastroPlus, Simcyp Simulator, Berkeley Madonna. |
| Human Physiological Database | Source for system parameters (organ volumes, blood flows, lymph flows, etc.). | ICRP Publications, PK-Sim Ontology. |
| Quantitative Proteomics Data | Resource for tissue-specific expression levels of FcRn and other relevant proteins. | The Human Protein Atlas, literature. |
Critical Differences Between Small Molecule and Large Molecule PBPK Modeling
This Application Note, within the broader thesis on PBPK for monoclonal antibodies (mAbs) and therapeutic proteins, delineates the fundamental distinctions in Physiologically-Based Pharmacokinetic (PBPK) modeling between small molecules and large molecules. These differences stem from disparate physicochemical properties and absorption, distribution, metabolism, and excretion (ADME) mechanisms, necessitating unique modeling frameworks.
Core Model Structure & Determinants of Disposition
Table 1: Fundamental Differences in PBPK Model Structure
| Aspect | Small Molecule PBPK | Large Molecule (mAb/Protein) PBPK |
|---|---|---|
| Primary Disposition Drivers | Passive diffusion, protein binding, metabolism by CYPs, transporter affinity. | Target-mediated drug disposition (TMDD), FcRn recycling, endocytotic clearance, immunogenicity. |
| Distribution | Typically rapid, described by tissue:plasma partition coefficients (Kp). | Typically slow, rate-limited by vascular permeability (vasculature reflection coefficient, σ) and convective flow; described by lymph flow and endocytic uptake. |
| Elimination | Hepatic metabolism (Km, Vmax), biliary excretion, renal filtration of unbound drug. | Linear/non-linear proteolytic catabolism in tissues, renal filtration followed by lysosomal degradation (for peptides), anti-drug antibody (ADA) clearance. |
| Binding | Non-specific plasma protein binding (e.g., to albumin). | Specific, high-affinity binding to target antigen (Kon, Koff), and protective binding to FcRn (Kd ~ µM range). |
| Critical Parameters | LogP, pKa, intrinsic clearance (CLint), fu. | Target antigen concentration (Rtotal), internalization rate (kint), FcRn affinity, endosomal pH, interstitial lymph flow. |
Experimental Protocols for Key Large Molecule-Specific Assays
Protocol 2.1: Determination of Target Antigen Concentration in Tissues Objective: Quantify total (membrane-bound + soluble) target antigen concentration for TMDD model parameterization. Materials: Homogenization buffer, protease inhibitors, validated ELISA kit (capture/detection antibodies for target), tissue homogenizer, microplate reader. Procedure:
- Weigh and homogenize tissue samples (e.g., tumor, liver) in ice-cold buffer with inhibitors.
- Centrifuge homogenate at 100,000 x g for 60 min at 4°C. Retain supernatant.
- Perform serial dilutions of supernatant and included standards on the ELISA plate.
- Incubate per kit instructions, develop, and read absorbance.
- Calculate tissue antigen concentration (e.g., in nmol/g tissue) using standard curve, correcting for dilution and tissue weight.
Protocol 2.2: In Vitro FcRn Binding Affinity Assay at Endosomal pH Objective: Measure the pH-dependent binding affinity (Kd) of mAb to human FcRn. Materials: Biacore or Octet system, recombinant human FcRn, test mAb, HBS-EP buffer, acetate buffer (pH 5.5), phosphate buffer (pH 7.4). Procedure (Biacore):
- Immobilize test mAb on a CMS sensor chip via amine coupling.
- Dilute FcRn in pH 5.5 buffer. Inject a concentration series (e.g., 0.1-10 µM) over the mAb surface at pH 5.5 (association phase).
- Switch to pH 7.4 buffer for the dissociation phase to mimic endosomal release.
- Regenerate the surface with a brief pH 2.0 buffer pulse.
- Analyze sensorgrams using a 1:1 binding model to derive Kon, Koff, and Kd at pH 5.5.
Visualizing Key Pathways & Model Structures
Title: mAb PBPK Core Pathways: FcRn Recycling & Target-Mediated Disposition
Title: PBPK Model Construction Workflow Comparison
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Large Molecule PBPK Model Parameterization
| Reagent/Material | Function in PBPK Context |
|---|---|
| Recombinant Human FcRn | For in vitro binding assays to determine the critical pH-dependent affinity parameter (Kd) governing recycling and half-life. |
| Target Antigen (Soluble & Membrane-Bound Forms) | Used to develop binding assays (SPR/BLI) to measure Kon/Koff for TMDD model, and as standards for tissue antigen quantification. |
| Anti-Drug Antibody (ADA) Positive Control Serum | To validate assays for ADA detection, a key input for modeling immunogenicity-driven clearance. |
| Tissue Homogenization Kits (Protease Inhibited) | For preparation of tissue lysates to quantify baseline target antigen expression levels (Rtotal) across organs. |
| Human/Monkey Tissue Sections (FFPE or Frozen) | For immunohistochemistry (IHC) to visualize and semi-quantify target and mAb distribution spatially, informing model structure. |
| pH-Gradient Chromatography Columns | To assess charge variants of mAbs, as isoelectric point (pI) influences capillary permeability and interstitial distribution. |
| Validated ELISA Kits for Soluble Targets/Biomarkers | To measure pharmacokinetic (PK) and pharmacodynamic (PD) biomarkers in vivo for model verification. |
| SPR (Biacore) or BLI (Octet) Biosensor Systems | Gold-standard platforms for obtaining quantitative binding kinetics (Kon, Koff, Kd) for mAb-antigen and mAb-FcRn interactions. |
Current Regulatory Landscape and Expectations for Biologics PBPK
Within the broader thesis on PBPK modeling for monoclonal antibodies (mAbs) and therapeutic proteins, understanding the current regulatory posture is essential. Regulatory agencies, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), recognize the potential of PBPK for biologics but underscore the need for rigorous, context-of-use specific model qualification. Unlike small-molecule PBPK, which is routinely used for drug-drug interaction (DDI) predictions, biologics PBPK is evolving from a research tool towards regulatory acceptance for specific applications like first-in-human (FIH) dose prediction, pediatric extrapolation, and predicting the impact of target-mediated drug disposition (TMDD) and immunogenicity.
Table 1: Key Regulatory Documents and Positions on Biologics PBPK
| Agency | Document/Guidance | Year | Relevant Position on Biologics PBPK |
|---|---|---|---|
| U.S. FDA | Physiologically Based Pharmacokinetic Analyses — Format and Content Guidance for Industry | 2022 | Encourages use for both small & large molecules; specifies data requirements for model validation, including system and drug-specific parameters. |
| EMA | Guideline on the reporting of physiologically based pharmacokinetic (PBPK) modelling and simulation | 2021 | Acknowledges utility for mAbs; emphasizes comprehensive sensitivity analysis and external validation. |
| U.S. FDA & EMA | Workshop Proceedings: PBPK Modeling for Biologics | 2023 (Workshop) | Highlighted priority use cases: FIH dosing, DDI with small molecules, and neonatal Fc receptor (FcRn) modulator interactions. Noted challenges in characterizing intracellular trafficking and anti-drug antibody (ADA) impact. |
Key Application Notes and Protocols
Application Note 1: FIH Dose Selection for a Novel Monoclonal Antibody
Objective: To predict safe and efficacious FIH dose ranges using a minimal PBPK (mPBPK) model incorporating TMDD.
Protocol: mPBPK-TMDD Model Development and FIH Simulation
Step 1: System Parameters.
- Use a validated mPBPK structure (2-3 tissue compartments). Standard human physiology parameters (organ volumes, blood flows, lymph flow, plasma volumes) are obtained from literature.
- Critical system parameter: FcRn-mediated recycling parameters (binding affinity, endosomal pH, FcRn concentration).
Step 2: Drug-Specific Parameterization.
- In vitro assays:
- Target Binding: Measure affinity (KD) to soluble and membrane-bound target using Surface Plasmon Resonance (SPR).
- FcRn Affinity: Determine pH-dependent binding kinetics at pH 6.0 and 7.4.
- Non-Specific Interactions: Estimate pinocytotic rate and endosomal degradation rate.
- In vivo (preclinical) studies in relevant animal model (e.g., humanized mouse, cynomolgus monkey):
- Conduct single-dose PK studies at sub-pharmacologic and pharmacologic doses to observe linear vs. nonlinear clearance.
- Fit data to estimate parameters for linear clearance (CL) and Michaelis-Menten constants (KM, Vmax) for target-mediated clearance.
Step 3: Model Construction & Verification.
- Integrate parameters into a differential equation-based mPBPK-TMDD model using platforms like GastroPlus, PK-Sim, or custom code in MATLAB/R.
- Verify the model by simulating preclinical PK data and comparing predictions to observed data (e.g., visual predictive checks, fold-error of AUC and Cmax within 2-fold).
Step 4: FIH Dose Prediction.
- Scale all system parameters to human values.
- Simulate a range of potential FIH doses (e.g., 0.1 mg/kg to 10 mg/kg).
- Output predictions: exposure (AUC, Cmax), target occupancy over time, and receptor turnover dynamics.
- Recommend a safe starting dose (typically 1/6th of the human equivalent dose from the no-observed-adverse-effect level (NOAEL) in animals) and project therapeutic dose ranges.
Title: Workflow for FIH Dose Prediction Using mPBPK
Application Note 2: Assessing DDI Risk Between a mAb and a Small Molete Drug
Objective: To evaluate if a therapeutic mAb, by modulating a cytokine target, can alter the cytochrome P450 (CYP) enzyme expression and affect the PK of a co-administered small molecule drug.
Protocol: Cytokine-Mediated DDI Assessment PBPK Protocol
Step 1: In Vitro Evidence Generation.
- Use primary human hepatocytes or relevant hepatocyte cell lines.
- Expose cells to the cytokine (target of the mAb) at pathophysiological concentrations.
- Measure changes in mRNA and/or activity of major CYP enzymes (e.g., CYP1A2, 2C9, 2C19, 2D6, 3A4) over 48-72 hours using qPCR and probe substrate assays.
Step 2: In Vivo Preclinical Confirmation (if feasible).
- Use an animal model with humanized target and relevant CYP orthologs.
- Administer the cytokine or an inducing agent, with/without the mAb.
- Measure PK of a sensitive CYP probe substrate (e.g., midazolam for CYP3A4).
Step 3: Integrated PBPK Modeling.
- Build a small-molecule PBPK model for the probe drug (e.g., midazolam).
- Build a systems pharmacology model for the cytokine-mAb interaction and its effect on CYP gene transcription/ degradation.
- Integrate the two models: Link the predicted cytokine suppression (by mAb) to the change in CYP enzyme abundance over time in the liver compartment of the small-molecule model.
- Qualify the model against any preclinical DDI data or clinical data from similar mAbs.
Step 4: Clinical DDI Prediction.
- Simulate the predicted PK of the probe drug with and without co-administration of the mAb at steady state.
- Calculate the geometric mean ratio (GMR) of AUC and Cmax.
- Apply regulatory thresholds (e.g., GMR 90% CI outside 0.8-1.25) to determine if a clinical DDI study is warranted.
Title: PBPK Workflow for Cytokine-Mediated mAb DDI Assessment
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Biologics PBPK Experimentation
| Category/Item | Function in PBPK Workflow | Example/Supplier |
|---|---|---|
| SPR/Biacore Systems | Gold-standard for measuring real-time kinetics of mAb binding to target antigen and FcRn at different pH levels. Critical for TMDD & FcRn parameterization. | Cytiva Biacore, Sartorius Octet |
| Human Hepatocytes (Primary or iPSC-derived) | In vitro system to assess cytokine-mediated regulation of CYP enzymes for DDI risk assessment. | Lonza, BioIVT, ReproCELL |
| Human FcRn Transgenic Mice | In vivo model with human FcRn expression for more predictive PK studies of mAbs, especially for FcRn-dependent recycling and half-life prediction. | GenOway, The Jackson Laboratory |
| PBPK Software with mAb Capabilities | Platforms enabling construction of multi-scale PBPK models for large molecules, featuring TMDD, FcRn, and lymph flow components. | Certara PK-Sim & MoBi, Simulations Plus GastroPlus, Open Systems Pharmacology Suite |
| Anti-Drug Antibody (ADA) Assay Kits | To quantify ADA incidence and titer in preclinical/clinical studies, enabling modeling of ADA impact on clearance and immunogenicity risk. | Meso Scale Discovery (MSD) Immunoassays, Gyros Protein Technologies |
| Recombinant Human Targets & FcRn | High-quality proteins for in vitro characterization of binding interactions, essential for accurate model input parameters. | ACROBiosystems, Sino Biological, R&D Systems |
From Theory to Practice: Building and Applying PBPK Models for Biologics
This application note provides a structured framework for developing a physiologically-based pharmacokinetic (PBPK) model for monoclonal antibodies (mAbs). Framed within a broader thesis on advancing PBPK for therapeutic proteins, this protocol aims to standardize the model development process for researchers and drug development professionals.
Model Framework and Core Structure
The foundational structure of a mAb PBPK model accounts for the unique pharmacokinetic properties of large molecules, including convection via lymphatic flow, target-mediated drug disposition (TMDD), and FcRn-mediated recycling.
Table 1: Core Physiological Parameters for a mAb PBPK Model
| Organ/Tissue | Volume (L, 70kg) | Plasma Flow Rate (L/h) | Lymph Flow Rate (L/h) | Vascular Reflection Coefficient (σ₁) | Lymphatic Reflection Coefficient (σ₂) |
|---|---|---|---|---|---|
| Plasma | 3.0 | - | - | - | - |
| Liver | 1.5 | 50.4 | 0.35 | 0.95 | 0.2 |
| Muscle | 30.0 | 30.0 | 0.10 | 0.95 | 0.2 |
| Skin | 3.3 | 9.6 | 0.15 | 0.95 | 0.2 |
| Gut | 1.4 | 36.0 | 0.30 | 0.95 | 0.2 |
| Heart | 0.35 | 14.4 | 0.02 | 0.95 | 0.2 |
| Kidney | 0.28 | 38.4 | 0.05 | 0.99 | 0.1 |
| Rest of Body | 10.17 | 71.2 | 0.43 | 0.95 | 0.2 |
Experimental Protocols for Model Parameterization
Protocol 2.1: Determining FcRn Association (Ka) and Dissociation (Kd) Constants via Surface Plasmon Resonance (SPR)
Objective: To quantify the binding affinity of the mAb to human FcRn at endosomal pH (6.0) and release pH (7.4).
Materials:
- Biacore T200 SPR system (Cytiva).
- CMS sensor chip.
- Recombinant human FcRn protein.
- Running Buffer: HBS-EP+ (10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.05% v/v Surfactant P20), pH-adjusted to 6.0 and 7.4.
- Regeneration Solution: 10 mM Glycine-HCl, pH 2.0.
Procedure:
- Chip Preparation: Immobilize recombinant human FcRn onto a CMS sensor chip via amine coupling to achieve ~5000 RU.
- Binding Kinetics at pH 6.0: Dilute the mAb in running buffer (pH 6.0). Inject a concentration series (e.g., 0, 3.125, 6.25, 12.5, 25, 50 nM) over the FcRn surface at a flow rate of 30 µL/min for 180s association time.
- Dissociation Phase: Monitor dissociation in pH 6.0 buffer for 300s.
- Regeneration: Inject regeneration solution for 30s to fully dissociate the complex.
- Repeat at pH 7.4: Repeat steps 2-4 using pH 7.4 running buffer.
- Data Analysis: Fit sensorgrams globally using a 1:1 Langmuir binding model in the Biacore Evaluation Software to derive Ka (association rate, 1/Ms), Kd (dissociation rate, 1/s), and KD (equilibrium dissociation constant, M).
Protocol 2.2: In Vivo Plasma PK Study for Model Calibration
Objective: To obtain concentration-time data for model fitting and validation.
Materials:
- Test mAb formulation.
- Animal model (e.g., human FcRn transgenic mouse, cynomolgus monkey).
- EDTA-coated blood collection tubes.
- Validated ELISA or LC-MS/MS assay for mAb quantification.
Procedure:
- Dosing: Administer the mAb intravenously at two distinct doses (e.g., 1 mg/kg and 10 mg/kg) to groups of animals (n=3-5 per dose).
- Serial Blood Sampling: Collect blood samples at pre-dose, 5 min, 4h, 12h, 24h, 3d, 7d, 14d, 21d, and 28d post-dose.
- Sample Processing: Centrifuge blood samples at 4°C, separate plasma, and store at -80°C until analysis.
- Bioanalysis: Quantify mAb concentrations in plasma using the validated assay.
- Data Compilation: Calculate mean (±SD) concentration at each time point for each dose group.
Table 2: Example In Vivo PK Data (Mean ± SD) for Model Input
| Time (h) | 1 mg/kg Concentration (µg/mL) | 10 mg/kg Concentration (µg/mL) |
|---|---|---|
| 0.083 | 14.2 ± 1.5 | 142.0 ± 15.2 |
| 24 | 8.1 ± 0.9 | 85.3 ± 9.1 |
| 168 | 1.2 ± 0.2 | 15.4 ± 2.1 |
| 336 | 0.15 ± 0.05 | 2.1 ± 0.4 |
Model Development and Workflow
Diagram Title: mAb PBPK Model Development Workflow
Key mAb Disposition Pathways
Diagram Title: Key Pathways in mAb PBPK Disposition
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for mAb PBPK Model Development
| Item/Category | Example Product/Source | Function in mAb PBPK Context |
|---|---|---|
| SPR System | Biacore Series (Cytiva) | Quantifies binding kinetics (Ka, Kd) of mAb to FcRn and soluble target antigens. |
| Recombinant Human FcRn | Sino Biological, Themo Fisher | Critical reagent for in vitro binding assays to parameterize the FcRn salvage mechanism. |
| Human FcRn Transgenic Mouse | B6.Cg-Fcgrt tm1Dcr Tg(FCGRT)32Dcr | In vivo model with humanized FcRn pathway for predictive preclinical PK studies. |
| PBPK Modeling Software | PK-Sim, Simcyp Simulator | Platform for implementing the mathematical model, performing simulations, and parameter estimation. |
| mAb Quantitation Assay | Gyrolab xPlore, ELISA | High-sensitivity, high-throughput bioanalytical method for generating PK concentration-time data. |
| Physiological Databases | ICRP Publications, literature | Source for human organ weights, blood flows, lymph flows, and vascular properties for model parameterization. |
Integrating Target-Mediated Drug Disposition (TMDD) and FcRn Mechanisms
Application Notes
The integration of Target-Mediated Drug Disposition (TMDD) and the neonatal Fc receptor (FcRn) salvage pathway is a critical advancement in the physiologically-based pharmacokinetic (PBPK) modeling of monoclonal antibodies (mAbs) and therapeutic proteins. This integrated framework is essential for accurately predicting the complex, non-linear PK observed for many biologics, where disposition is simultaneously influenced by saturable target binding and concentration-dependent FcRn-mediated recycling.
Core Conceptual Integration: In a typical mAb PBPK model, the antibody is distributed via vascular and lymphatic flow. The integrated model must account for two primary clearance/saturation mechanisms:
- FcRn Mechanism: Provides the baseline linear-to-saturating PK. At low concentrations, FcRn in vascular endothelium and hematopoietic cells binds to the Fc region of IgG in acidic endosomes, recycling it back to the systemic circulation and preventing lysosomal degradation. This process is saturable at high antibody concentrations.
- TMDD Mechanism: Introduces target-specific non-linearity. The antibody binds with high affinity to its soluble or membrane-bound target. The resulting complex may undergo internalization and degradation, constituting an additional, often saturable, clearance pathway.
The interplay between these systems dictates overall PK. For instance, a mAb with high target affinity may show pronounced TMDD at low doses, while FcRn saturation may dominate at very high doses. The integrated model quantitatively dissects these contributions, which is vital for optimal first-in-human dosing, dose regimen selection, and extrapolation from preclinical species.
Key Applications in Drug Development:
- First-in-Human Dose Prediction: De-risking clinical translation by separating target-mediated clearance from non-specific clearance.
- Optimal Dosing Regimen Design: Informing loading and maintenance dose strategies to achieve target engagement while minimizing clearance.
- Interspecies Scaling: Facilitating human PK prediction from preclinical data by incorporating species-specific FcRn affinity and target expression levels.
- Special Population Simulations: Predicting PK alterations in populations with modulated target burden (e.g., inflammatory disease states) or altered FcRn expression (e.g., pregnant patients).
Quantitative Data Summary:
Table 1: Key Model Parameters for Integrated TMDD-FcRn PBPK Models
| Parameter Category | Symbol | Typical Value Range (Human) | Description & Impact |
|---|---|---|---|
| FcRn Parameters | K_FcRn |
100 - 600 nM | Affinity of mAb for FcRn at acidic pH (~6.0). Lower affinity reduces recycling, increasing clearance. |
FcRn_max |
Tissue-specific (e.g., ~0.5 µM in endothelium) | Maximum FcRn concentration in relevant tissues. Limits recycling capacity. | |
| TMDD Parameters | K_on, K_off |
e.g., 10^5 M⁻¹s⁻¹, 10^-4 s⁻¹ | Association/dissociation rate constants for mAb-target binding. |
K_D (=K_off/K_on) |
pM to nM | Equilibrium dissociation constant. Lower K_D indicates higher target affinity. | |
K_int |
0.1 - 5 day⁻¹ | Internalization rate constant of mAb-target complex. Major driver of TMDD clearance. | |
| Target Parameters | R_total |
pg/mg - ng/mg tissue | Baseline total target expression level. Critical for scaling from animals to humans. |
k_syn, k_deg |
e.g., 0.1 - 10 pmol/L/day | Target synthesis and degradation rates. Determines target turnover. |
Table 2: Example PK Outcomes from Model Simulations
| Scenario (Dose Level) | Dominant Mechanism | Observed PK Profile | Clinical Development Implication |
|---|---|---|---|
| Very Low Dose | TMDD | Highly non-linear; rapid clearance. | Sub-therapeutic exposure likely. Avoid this range. |
| Therapeutic Dose (Low) | Mixed (TMDD > FcRn) | Non-linear; clearance decreases with dose. | Dose increases yield more-than-proportional exposure gains. |
| Therapeutic Dose (High) | Mixed (FcRn ≥ TMDD) | Approaching linearity. | Predictable, dose-proportional PK. |
| Very High Dose | FcRn Saturation | Non-linear; clearance increases as FcRn saturated. | Potential for increased clearance and waste of drug. |
Experimental Protocols
Protocol 1:In VitroDetermination of FcRn Binding Affinity (Surface Plasmon Resonance)
Objective: To measure the affinity (K_D) of the mAb for human FcRn at pH 6.0.
Methodology:
- Immobilization: Dilute biotinylated human FcRn to 5 µg/mL in HBS-EP+ buffer (pH 7.4). Inject over a streptavidin (SA) sensor chip for 7 minutes to achieve ~1000 RU immobilization level.
- Kinetic Run: Using a SPR instrument (e.g., Biacore), perform kinetic measurements at 25°C. Use running buffer: 50 mM MES, 150 mM NaCl, 0.05% P20, pH 6.0.
- Sample Injection: Inject a 2-fold serial dilution of the mAb (e.g., 500 nM to 3.9 nM) over the FcRn and reference flow cells for 3 minutes (association), followed by dissociation for 10 minutes in pH 6.0 buffer.
- Regeneration: Regenerate the surface with two 30-second pulses of HBS-EP+ buffer (pH 7.4).
- Data Analysis: Double-reference the data (reference flow cell and zero-concentration blank). Fit the association and dissociation phases globally to a 1:1 Langmuir binding model using the instrument's evaluation software to obtain
K_on,K_off, andK_D.
Protocol 2:In VitroCellular Assay for TMDD Parameters (Internalization Rate)
Objective: To determine the internalization rate constant (k_int) of the mAb-target complex using a cell line expressing the target.
Methodology:
- Cell Preparation: Seed cells expressing the membrane target (e.g., HEK293 overexpressing Target X) in a 24-well plate at 2.5 x 10^5 cells/well. Culture overnight.
- Surface Binding (4°C): Cool plates on ice. Wash cells with cold assay buffer. Add a saturating concentration of fluorescently-labeled mAb (e.g., 100 nM) in cold buffer. Incubate for 1 hour on ice to allow binding without internalization.
- Internalization Phase (37°C): Wash cells thoroughly with cold buffer to remove unbound mAb. Add pre-warmed serum-free medium and immediately transfer plates to a 37°C, 5% CO₂ incubator. Incubate for various time points (t= 2, 5, 15, 30, 60, 120 min).
- Acid Stripping: At each time point, place the plate on ice. Remove medium and wash cells with cold buffer. Treat cells with an acidic strip buffer (e.g., 0.2M acetic acid, 0.5M NaCl, pH 2.5) for 5 minutes on ice to remove antibody remaining on the cell surface.
- Lysate Preparation: Neutralize the acid, wash cells, and lyse them in RIPA buffer. Measure the fluorescence intensity of the lysate (representing internalized mAb) using a plate reader.
- Data Analysis: Plot internalized fluorescence vs. time. Fit the initial linear phase (typically up to 30 min) to the equation:
Internalized Signal = k_int * [Surface Bound] * t. The slope provides an estimate ofk_int.
Protocol 3:In VivoPK Study for Model Validation
Objective: To generate PK data in humanized FcRn transgenic mice for integrated model validation. Methodology:
- Animal Dosing: Use homozygous human FcRn transgenic mice (e.g., B6.Cg-Fcgrttm1Dcr Tg(FCGRT)32Dcr/DcrJ). Administer the mAb via a single intravenous bolus injection at three distinct dose levels (e.g., 0.5 mg/kg, 5 mg/kg, and 50 mg/kg) to capture both TMDD and FcRn saturation phases (n=4-6 per group).
- Serial Blood Sampling: Collect blood samples (~25 µL) via a validated microsampling technique (e.g., tail vein) at pre-dose, 0.083, 0.5, 1, 3, 7, 24, 48, 96, 168, 240, and 336 hours post-dose.
- Bioanalysis: Process plasma by solid-phase extraction. Quantify mAb concentrations using a validated target-capture ELISA (to measure free mAb) and a total mAb ELISA.
- PK Analysis & Modeling: Perform non-compartmental analysis (NCA) to estimate AUC and clearance. Subsequently, fit the concentration-time data from all dose levels simultaneously using the integrated TMDD-FcRn PBPK model in a software platform (e.g., GastroPlus, Simbiology, or custom differential equations in R). Estimate key in vivo parameters (
K_int,R_total,FcRn_max) and validate model predictive performance.
Visualizations
Diagram Title: Integrated TMDD and FcRn Pathways for mAb PK
Diagram Title: Workflow for Developing Integrated TMDD-FcRn PBPK Model
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Key Reagents and Materials for Integrated TMDD-FcRn Research
| Item | Function in Research | Example/Supplier |
|---|---|---|
| Recombinant Human FcRn Protein | Critical for in vitro binding affinity assays (SPR, ELISA) to determine K_D at acidic pH. |
AcroBiosystems, Sino Biological, R&D Systems. |
| Human FcRn Transgenic Mouse Model | In vivo model with human-like IgG/FcRn interaction kinetics for predictive PK studies. | B6.Cg-Fcgrttm1Dcr Tg(FCGRT)32Dcr (Jackson Lab). |
| Cell Line Overexpressing Target Antigen | Required for cellular internalization (k_int) assays and in vitro potency assessments. |
Generated in-house or from repositories like ATCC. |
| pH-Sensitive Assay Buffers | For mimicking endosomal (pH 6.0) and physiological (pH 7.4) conditions in FcRn binding studies. | MES (pH 6.0), PBS or HEPES (pH 7.4). |
| Anti-Idiotypic Antibodies | Reagents for developing drug-specific PK ELISAs (total and free assay formats). | Generated via custom immunization (e.g., Abzena, GenScript). |
| PBPK/PD Modeling Software | Platform for building, simulating, and fitting the integrated mechanistic model. | Simbiology (MATLAB), GastroPlus, Berkeley Madonna, R/PKPDsim. |
| Surface Plasmon Resonance (SPR) Instrument | Gold-standard for label-free, real-time measurement of binding kinetics (K_on, K_off). |
Biacore (Cytiva), Sierra SPR (Bruker). |
| Microsampling Equipment | Enables serial blood sampling from a single mouse, improving data quality and reducing animal use. | EDTA-coated capillaries, Mitra devices (Neoteryx). |
Application in First-in-Human (FIH) Dose Prediction and Scaling
Within the broader thesis on the application of Physiologically-Based Pharmacokinetic (PBPK) modeling for monoclonal antibodies (mAbs) and therapeutic proteins, the prediction of the safe and efficacious First-in-Human (FIH) dose represents a critical translational milestone. This document outlines the application notes and protocols for integrating in vitro and in silico data to predict human pharmacokinetics (PK) and pharmacodynamics (PD), thereby enabling rational FIH dose selection and scaling.
The following tables consolidate key quantitative parameters utilized in FIH dose prediction for mAbs and therapeutic proteins.
Table 1: Key In Vitro to In Vivo Scaling Parameters for mAbs
| Parameter | Symbol | Typical Value Range (Human) | Source/Determination Method | Purpose in FIH Scaling |
|---|---|---|---|---|
| Plasma Clearance | CL | 0.2 - 0.5 L/day for IgG1 | Allometric scaling from preclinical species (exponent ~0.8-0.9) | Predicts human systemic exposure (AUC) |
| Volume of Distribution at Steady State | Vss | 3.5 - 5.5 L | Correlates with plasma volume; scaling via fixed exponent (~1.0) | Predicts peak (Cmax) and trough concentrations |
| Target Affinity (Dissociation Constant) | Kd | pM - nM range | Surface Plasmon Resonance (SPR) | Informs target engagement and PK/PD model |
| Linear Elimination Half-life | t1/2 | 14 - 21 days | Derived from CL and Vss (t1/2 = 0.693*Vss/CL) | Dosing interval determination |
| Neonatal Fc Receptor (FcRn) Affinity (pH 6.0) | - | KD ~ 300-600 nM | In vitro FcRn binding assay | Predicts recycling and half-life |
Table 2: Common Safety Margins and Starting Dose Criteria
| Approach | Calculation Basis | Typical Safety Margin (Multiples) | Application Context |
|---|---|---|---|
| Minimum Anticipated Biological Effect Level (MABEL) | In vitro IC/EC50 for pharmacological effect | 10x - 100x below predicted pharmacologically active dose | High-risk candidates (e.g., super-agonists, novel targets) |
| No Observed Adverse Effect Level (NOAEL) | Highest dose from GLP toxicology studies | 1/10th of human equivalent NOAEL (based on AUC or dose) | Standard mAbs with clean toxicology profile |
| Pharmacologically Active Dose (PAD) | Integrated PK/PD modeling from preclinical data | Starting dose often a fraction (e.g., 1/10th) of PAD | When robust in vivo efficacy data exists |
Experimental Protocols
Protocol 1:In VitroFcRn Binding Affinity Assay for Half-life Prediction
Purpose: To determine the binding affinity of a mAb to human FcRn at endosomal pH (6.0) as a key parameter for PBPK model input to predict human clearance. Materials: See "Scientist's Toolkit" (Section 5). Procedure:
- Biosensor Preparation: Immobilize recombinant human FcRn onto a CMS sensor chip via amine coupling to achieve ~1000 Response Units (RU).
- Running Buffer Preparation: Prepare HBS-EP+ buffer (10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.05% v/v Surfactant P20), pH-adjusted to 6.0 and 7.4.
- Kinetic Analysis: a. Dilute the mAb analyte in pH 6.0 buffer to a concentration series (e.g., 0, 12.5, 25, 50, 100, 200 nM). b. Using a Biacore/SPR system, inject each analyte concentration over the FcRn surface at a flow rate of 30 µL/min for an association phase of 120 seconds. c. Switch to pH 7.4 running buffer for a 300-second dissociation phase. d. Regenerate the surface with a 30-second pulse of pH 7.4 buffer containing 1 M NaCl.
- Data Analysis: Fit the resulting sensorgrams globally to a 1:1 Langmuir binding model using the evaluation software. Report the association rate (ka), dissociation rate (kd), and calculated equilibrium dissociation constant (KD = kd/ka).
Protocol 2: Allometric Scaling of Clearance from Preclinical Species
Purpose: To predict human clearance (CL) using simple allometric scaling from in vivo PK studies in multiple species. Materials: PK data (CL values) from at least three preclinical species (e.g., mouse, rat, monkey). Procedure:
- Data Compilation: Tabulate the measured clearance (CL, in mL/day) and average body weight (BW, in kg) for each species.
- Allometric Equation: Apply the power law: CL = a * BW^b. Perform a log-transformation: log(CL) = log(a) + b * log(BW).
- Linear Regression: Perform a simple linear regression of log(CL) against log(BW) to obtain the intercept (log(a)) and the allometric exponent (b).
- Human Prediction: For a standard human body weight of 70 kg, calculate the predicted human clearance: Predicted CLhuman = a * (70)^b.
- Incorporation into PBPK: Use the predicted CLhuman as an initial input parameter for the systemic clearance in the whole-body PBPK model.
Visualizations
Title: PBPK-Based Workflow for FIH Dose Prediction
Title: FcRn-Mediated mAb Recycling Pathway
The Scientist's Toolkit
Table 3: Key Research Reagent Solutions for FIH Prediction Assays
| Item | Function in FIH Prediction | Example/Supplier Note |
|---|---|---|
| Recombinant Human FcRn Protein | Critical for in vitro binding assays to predict antibody half-life. | Produced in HEK293 or CHO cells; available from multiple biotech suppliers (e.g., Sino Biological, Acro Biosystems). |
| SPR/Biacore Instrumentation | Gold-standard for label-free kinetic analysis of protein-protein interactions (e.g., mAb-FcRn, mAb-target). | Systems from Cytiva (Biacore) or Bruker (BLAcore). |
| Species-Specific Serum/Plasma | Used in in vitro stability and protein-binding studies to inform clearance. | Pooled, gender-matched, commercially available from vendors like BioIVT or SeraCare. |
| PBPK Modeling Software Platform | Enables integration of in vitro and in vivo data for human PK prediction. | Commercial: Simcyp Simulator, GastroPlus. Open-source: PK-Sim. |
| Immunoassay Kits (ELISA/MSD) | Quantification of mAb/therapeutic protein concentrations in preclinical PK studies. | Requires target- or drug-specific reagents. MSD plates offer high sensitivity. |
| In Vivo PK Study Materials (Preclinical) | Conducting PK studies in relevant species (mouse, rat, NHP) to generate scaling data. | Includes dosing formulations, catheters for serial sampling, and appropriate animal models. |
Within the broader thesis on PBPK modeling for monoclonal antibodies (mAbs) and therapeutic proteins, the application to special populations represents a critical advancement. Traditional clinical trials often exclude pediatric, pregnant, or organ-impaired patients, creating significant knowledge gaps. PBPK modeling, integrating drug-specific properties with population-specific physiology, provides a mechanistic framework to predict pharmacokinetics (PK) in these groups, optimizing dosing and de-risking development.
Quantitative Data Summaries
Table 1: Key Physiological Parameters for Special Populations in PBPK
| Physiological Parameter | Pediatrics (vs. Adult) | Pregnancy (vs. Non-Pregnant) | Hepatic Impairment (Child-Pugh B) |
|---|---|---|---|
| Cardiac Output | Higher per kg body weight | Increases up to 40-50% | Generally unchanged |
| Glomerular Filtration Rate (GFR) | Matures by 1 year; lower in neonates | Increases 40-50% by 2nd trimester | Decreased (moderate-severe) |
| Hepatic CYP450 Activity | Ontogeny profiles vary by enzyme | Variable (some increased) | Significantly decreased |
| Plasma Volume | Higher as % of body weight | Increases ~45% | May be increased (ascites) |
| Serum Albumin | Lower in neonates | Decreased by ~10-15% | Often decreased |
| Body Fat % | Variable with age | Increased | Variable |
Table 2: Example PBPK-Predicted vs. Observed PK Changes for Representative mAbs
| Drug/Therapeutic Protein | Population | Predicted Change in AUC (vs. Reference) | Observed Change (Literature) | Primary Physiological Driver in Model |
|---|---|---|---|---|
| Trastuzumab | Pediatrics (12y) | ~20% lower (scaled by weight/BSA) | ~25% lower | Body size, FcRn expression |
| Adalimumab (anti-TNFα) | Pregnancy (3rd Trimester) | ~30-40% lower | ~25-50% lower (cord blood ~1:1) | Increased volume, GFR, catabolic rate |
| Atezolizumab (anti-PD-L1) | Renal Impairment (Severe) | ~10% increase (limited impact) | Minimal change | Non-renal clearance dominant |
| Pegfilgrastim | Hepatic Impairment (Moderate) | Negligible change | No significant change | Neutrophil-mediated clearance |
Application Notes & Protocols
Protocol: Building a Pediatric PBPK Model for a Monoclonal Antibody
Objective: To develop a pediatric PBPK model for a novel IgG1 mAb from an established adult model.
Workflow Diagram:
Detailed Methodology:
- Base Model: Start with a verified whole-body PBPK model for the mAb in adults, featuring key mAb processes: convection via lymphatics, tissue distribution via permeability-surface area/endosomal trafficking, linear/nonlinear FcRn-mediated recycling, and target-mediated drug disposition (TMDD).
- Age Stratification: Define pediatric subgroups (e.g., 0-1 month, 1-24 months, 2-12 years, 12-18 years).
- Physiological Database: Use published resources (e.g., FDA's Pediatric PopPK Guidance, ICRP publications, PK-Sim Ontogeny Database) to populate model parameters:
- Organ volumes and blood flows: Scale using allometric principles (weight^0.75 for flows, weight^1 for volumes) with age-dependent coefficients.
- Plasma protein levels: Incorporate ontogeny of albumin and FcRn concentration. FcRn expression may be assumed at adult levels post-neonatal period.
- Lymphatic flow: Scale based on body size.
- GFR and organ function: Apply maturation functions (e.g., Hill equation) for renal filtration and hepatic metabolic enzymes if relevant for protein catabolism.
- Parameter Scaling: Systematically replace adult physiological values in the model with the pediatric counterparts for each age band.
- Sensitivity Analysis: Perform local or global sensitivity analysis to identify the most influential physiological parameters (e.g., GFR, FcRn concentration, cardiac output).
- Simulation & Validation: Simulate typical dosing regimens. Compare PK predictions (e.g., trough levels, AUC) against any available clinical data in pediatric populations for the same or similar mAbs. Visually and statistically (e.g., fold-error) assess concordance.
- Dose Optimization: Use the verified model to simulate alternative dosing (weight-based, BSA-based, fixed) to achieve exposure targets matching adult therapeutic levels.
Protocol: Simulating mAb PK in Pregnancy
Objective: To predict the exposure of a therapeutic IgG across trimesters and fetal transfer.
Workflow Diagram:
Detailed Methodology:
- Baseline Model: Use a female PBPK model (non-pregnant).
- Maternal Physiology: Modify parameters progressively by trimester based on literature:
- Increase: Plasma volume (up to 45%), cardiac output (up to 50%), GFR (up to 50%), body fat.
- Decrease: Serum albumin, some CYP activity (less relevant for mAbs).
- Add/Expand: Uterus, placenta, mammary tissue compartments with respective blood flows.
- Fetal Model: Add a fetal compartment, typically represented as a separate PBPK model (simplified) or as part of a linked "mother-placenta-fetus" system.
- Placental Transfer: Model the transfer of IgG using an FcRn-mediated transcytosis process across the placental barrier. This can be represented as a permeability-surface area coefficient or a dedicated kinetic process mirroring cellular recycling.
- Simulation: Run simulations across gestational ages. Key outputs include maternal PK profiles, time to fetal steady-state, and maternal-to-fetal concentration ratios (cord blood at delivery).
- Validation: Compare predicted ratios (often ~1:1 at term for IgG1) and temporal profiles with empirical data from other mAbs (e.g., infliximab, adalimumab).
Protocol: PBPK Modeling for mAbs in Renal or Hepatic Impairment
Objective: To assess the impact of chronic kidney disease (CKD) or liver cirrhosis on mAb PK.
Pathophysiology & Modeling Adjustments Diagram:
Detailed Methodology for Renal Impairment:
- Identify Clearance Pathways: Determine the contribution of renal elimination (glomerular filtration of fragments or intact protein) to total clearance from human ADME studies.
- Modify Model: If renal clearance is significant, correlate GFR (e.g., CKD-EPI equation) with the renal clearance parameter in the model. For severe CKD, also consider potential changes in fluid balance (edema), serum protein levels, and possibly altered FcRn expression in vascular endothelium.
- Simulation: Simulate PK profiles for subjects with mild, moderate, and severe renal impairment (e.g., CKD stages 2-5).
- Interpretation: For most intact mAbs, renal impairment shows minimal effect unless the mAb is small (e.g., Fab fragments, peptides). The model should confirm or quantify this.
Detailed Methodology for Hepatic Impairment:
- Identify Clearance Pathways: Hepatic impairment may affect mAb PK via changes in: (a) FcRn expression/function in sinusoidal endothelium, (b) blood flow affecting distribution, (c) impaired catabolism due to reduced proteolytic capacity, (d) altered target expression, and (e) increased gamma globulin levels.
- Modify Model: Adjust liver volume, hepatic blood flow (based on Child-Pugh score), serum albumin, and potentially reduce intrinsic catabolic rate in hepatic tissue. Changes in FcRn binding affinity or concentration can be tested if data suggests.
- Simulation & Validation: Simulate PK across Child-Pugh classes A, B, and C. Compare outcomes with available clinical data, noting that effects are often modest for mAbs.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Tools for Developing PBPK Models in Special Populations
| Tool/Resource Category | Specific Example/Name | Function & Relevance |
|---|---|---|
| PBPK Software Platform | PK-Sim (Open Systems Pharmacology), Simcyp Simulator, GastroPlus | Provides quantitative systems pharmacology (QSP) frameworks with built-in, verified population libraries for pediatrics, pregnancy, and disease states. Essential for simulation. |
| Physiological Databases | FDA Pediatric Guidance Documents, ICRP Publication 89, PopGen Pediatric Virtual Population | Sources for age-dependent organ weights, blood flows, enzyme ontogeny, and other system parameters needed to parameterize models. |
| Clinical PK Data Repositories | ClinicalTrials.gov, PubMed, Drug Approval Packages (FDA/EMA) | Sources of observed PK data in special populations for model validation. Critical for verifying predictions. |
| Biomarker Assay Kits | ELISA/MS kits for FcRn, target antigen, anti-drug antibodies (ADA) | Used to generate in vitro or ex vivo data (e.g., FcRn binding affinity, target concentration in disease) to inform model parameters. |
| In Silico Proteomics Tools | QSAR models for mAb tissue partition coefficients, in vitro-in vivo extrapolation (IVIVE) of clearance | Helps estimate drug-specific parameters when empirical data is lacking, especially for novel protein formats. |
| Statistical & Modeling Tools | R (with 'mrgsolve', 'PopED'), MATLAB/SimBiology, NONMEM | Used for model coding (if building from scratch), parameter estimation, sensitivity analysis, and population (PopPK) integration. |
Introduction & Thesis Context Physiologically-based pharmacokinetic (PBPK) modeling has evolved beyond small molecules to become a cornerstone in the development of monoclonal antibodies (mAbs) and therapeutic proteins. Within the broader thesis of advancing PBPK for large molecules, this article presents detailed application notes and protocols from three critical therapeutic areas. The framework integrates target-mediated drug disposition (TMDD), FcRn recycling, and tissue-scale dynamics to optimize dosing, predict drug-drug interactions, and support regulatory submissions.
Application Note 1: Oncology – Predicting Tumor Penetration for a PD-1 Inhibitor
Objective: To develop a whole-body PBPK model for a PD-1 inhibitor (pembrolizumab analog) to simulate its distribution into solid tumors (non-small cell lung cancer) and inform first-in-human (FIH) dosing.
Key Quantitative Data Summary: Table 1: Model Parameters and Simulation Outcomes for the Oncology mAb PBPK Model
| Parameter Category | Specific Parameter | Value (Mean) | Source/Justification |
|---|---|---|---|
| Systemic PK | Clearance (CL) | 0.22 L/day | Population PK analysis of clinical data |
| Central Volume (Vc) | 3.1 L | Allometric scaling from primates | |
| FcRn Affinity (KD) | 50 nM | In vitro surface plasmon resonance | |
| Tumor Physiology | Tumor Blood Flow Fraction | 1% of cardiac output | Literature data for NSCLC |
| Tumor Vascular Permeability (PS) | 3.0 x 10⁻⁸ cm/s | In vivo imaging study in xenografts | |
| Target Expression (PD-1) | 0.5 μM | Tumor biopsy IHC quantification | |
| Simulation Output | Predicted Trough Conc. at Steady State (2 mg/kg Q3W) | 45 μg/mL | PBPK model simulation |
| Predicted Tumor:Plasma AUC Ratio | 0.25 | PBPK model simulation | |
| Recommended Phase 2 Dose (RP2D) | 2 mg/kg Q3W | Integrated with PD biomarker data |
Detailed Protocol: PBPK Model Development and Tumor Penetration Simulation
1. In Vitro Assay for FcRn Binding Affinity
- Objective: Determine pH-dependent binding to human FcRn.
- Materials: Biacore T200 SPR system, CMS sensor chip, recombinant human FcRn, mAb analyte in buffers at pH 6.0 and 7.4.
- Procedure:
- Immobilize anti-Fc capture antibody on CMS chip via amine coupling.
- Dilute FcRn to 5 μg/mL in HBS-EP+ buffer (pH 6.0). Capture FcRn on flow cell.
- Inject mAb samples (0.5-500 nM) over FcRn surface at pH 6.0 for 180s association, followed by dissociation at pH 7.4 for 300s.
- Regenerate surface with Glycine-HCl, pH 2.0.
- Fit sensorgrams using a 1:1 Langmuir binding model to derive KD at pH 6.0.
2. Ex Vivo Tissue Partitioning via Cryo-imaging
- Objective: Quantify mAb distribution in tumor and normal tissues.
- Materials: Human tumor xenograft mouse model, fluorescently labeled mAb, cryo-microtome, fluorescence imaging system.
- Procedure:
- Administer 10 mg/kg fluorescent mAb IV to tumor-bearing mice (n=5).
- Euthanize at 24, 72, and 168h post-dose. Excise tumors, liver, muscle.
- Embed tissues in OCT, freeze, and section (50 μm thickness).
- Acquire high-resolution fluorescence images. Quantify intensity per tissue volume using calibration standards.
- Convert fluorescence to mAb concentration for PBPK model verification.
3. PBPK Model Simulation Workflow 1. Build base mAb PBPK model (two-pore formalism) in software (e.g., Simbiology, PK-Sim). 2. Populate system parameters (organ volumes, blood flows, lymph flows) from human physiology literature. 3. Incorporate drug-specific parameters: CL, Vc, FcRn KD, tissue-specific PS from in vitro/vivo studies. 4. Add tumor compartment: define volume growth rate, vascular surface area, and interstitial pressure. 5. Implement TMDD kinetics for PD-1 binding in tumor and peripheral lymphoid organs. 6. Validate model against clinical Phase I PK data. 7. Run simulations for proposed dosing regimens (1, 2, 5 mg/kg Q3W). Output: plasma PK, tumor interstitial concentration, receptor occupancy over time.
The Scientist's Toolkit: Key Research Reagent Solutions
- Recombinant Human FcRn Protein: For in vitro binding assays to characterize mAb half-life potential.
- Anti-Human PD-1 IHC Antibody (Validated): For quantifying target expression density in tumor biopsies for model input.
- Fluorescent Dye Labeling Kit (e.g., Alexa Fluor 647 NHS Ester): For preparing tracer antibody for in vivo and ex vivo distribution studies.
- PBPK Modeling Software (e.g., GastroPlus, Simbiology): Platform for integrating physiological, drug, and disease parameters.
PBPK Model Development Workflow for mAbs
Application Note 2: Immunology – Optimizing Dosing for an IL-17A Inhibitor in Psoriasis
Objective: To apply a PBPK-PD model incorporating IL-17A pathway kinetics to simulate optimal loading and maintenance dosing for a novel IL-17A inhibitor in psoriatic patients.
Key Quantitative Data Summary: Table 2: Key Parameters for the IL-17A Inhibitor PBPK-PD Model
| Parameter Category | Specific Parameter | Value | Note |
|---|---|---|---|
| Drug Parameters | Target Binding KD (IL-17A) | 0.1 nM | Cell-based bioassay |
| Non-Specific Clearance | 0.15 L/day | Estimated from preclinical species | |
| Disease Parameters | Psoriatic Plaque Blood Flow | 2x Normal Skin | Laser Doppler imaging data |
| IL-17A Production Rate in Plaque | 5 ng/day | Estimated from cytokine measurements | |
| Target (IL-17R) Expression | 10,000 receptors/cell | Flow cytometry on patient T-cells | |
| PD Biomarker | PASI Score (Baseline) | 15 (mean) | Clinical trial baseline |
| Simulation Output | Target IC90 for PASI75 | >85% RO at week 12 | Model correlation |
| Recommended Loading Dose | 400 mg SC | To achieve >90% RO in plaque by Week 2 | |
| Predicted PASI75 at Week 12 (Maintenance) | 72% | For 200 mg Q4W regimen |
Detailed Protocol: Integrating Cytokine Dynamics and Pharmacodynamics
1. Cell-Based Bioassay for IL-17A Neutralization
- Objective: Determine potency (IC50) of the therapeutic mAb.
- Materials: HEK-293 cells transfected with IL-17R/NF-κB reporter, recombinant human IL-17A, assay medium, luciferase detection kit.
- Procedure:
- Seed reporter cells in 96-well plates.
- Pre-incubate serial dilutions of the mAb (0.001-100 nM) with a fixed EC80 concentration of IL-17A (2 ng/mL) for 1 hour.
- Add mAb/cytokine mix to cells. Incubate for 24 hours.
- Lyse cells and measure luciferase activity. Fit dose-response curve to calculate IC50 and KD.
2. PBPK-PD Model Linking Skin Compartment to PASI Score 1. Develop whole-body PBPK model for a subcutaneous mAb, including a dedicated "psoriatic plaque" compartment with enhanced lymph flow and vascular permeability. 2. Implement TMDD kinetics for IL-17A binding in the plaque compartment. Define synthesis and degradation rates of free IL-17A based on literature. 3. Link the model to a downstream PD effect compartment (e.g., keratinocyte activation). 4. Establish an indirect response model where the inhibition of IL-17A signaling reduces the production rate of the PASI score. 5. Calibrate the model using Phase I PK data and Phase II PASI score time courses. 6. Simulate various loading/maintenance regimens. Identify dosing that achieves >90% receptor occupancy in plaque rapidly and sustains >80% for the dosing interval.
The Scientist's Toolkit: Key Research Reagent Solutions
- IL-17A Responsive Reporter Cell Line: For consistent, quantitative measurement of mAb neutralizing potency.
- Recombinant Human IL-17A Cytokine: Critical ligand for competitive binding assays and cell-based assays.
- Human Skin Explant Culture System: To study mAb penetration and local cytokine modulation ex vivo.
- PASI Scoring Guide & Digital Assessment Tool: For quantitative correlation of clinical PD endpoint with model predictions.
IL-17A Inhibition Pathway in Psoriatic Plaque
Application Note 3: Rare Disease – Pediatric Dose Selection for a Lysosomal Enzyme
Objective: To use a PBPK model incorporating age-dependent physiology and target expression (enzyme substrate) to extrapolate adult dosing of a recombinant lysosomal enzyme (e.g., for Gaucher disease) to pediatric populations.
Key Quantitative Data Summary: Table 3: Age-Dependent Parameters for Pediatric Enzyme PBPK Model
| Physiological Parameter | Neonate (3kg) | 5-Year-Old (18kg) | Adult (70kg) | Source |
|---|---|---|---|---|
| Body Weight (kg) | 3.0 | 18.0 | 70.0 | Standard growth charts |
| Plasma Volume (L) | 0.12 | 0.66 | 3.00 | Allometric scaling (BW^1.0) |
| Lymph Flow (L/day) | 1.5 | 6.8 | 18.0 | Allometric scaling (BW^0.75) |
| Tissue Mannose Receptor Expression (Liver) | 150% of adult | 120% of adult | 100% (Baseline) | Pediatric biopsy analysis* |
| Simulated Clearance (L/day) | 0.08 | 0.32 | 0.85 | PBPK model output |
| Model-Predicted Pediatric Dose | 1.5 mg/kg Q2W | 1.2 mg/kg Q2W | 1.0 mg/kg Q2W | To match adult exposure (AUC) |
*Estimated from limited data.
Detailed Protocol: Pediatric Physiological Scaling and Dose Rationale
1. Determination of Mannose Receptor Density
- Objective: Estimate age-dependent expression of the clearance receptor (CI-MPR) in liver.
- Materials: Banked pediatric and adult liver tissue samples, anti-CI-MPR antibody, quantitative Western blot or LC-MS/MS proteomics setup.
- Procedure:
- Homogenize tissue samples. Isolate membrane protein fraction.
- For Western blot: Separate proteins, transfer, probe with anti-CI-MPR and a reference protein (e.g., Na+/K+ ATPase). Quantify band intensity.
- For proteomics: Digest proteins, perform SRM/MRM mass spectrometry targeting CI-MPR peptides.
- Normalize receptor abundance to total protein or tissue weight. Express as relative ratio to adult mean.
2. Pediatric PBPK Simulation Protocol 1. Start with validated adult enzyme PBPK model incorporating TMDD via liver CI-MPR. 2. Scale all physiological compartments (organ volumes, blood flows, lymph flows) for pediatric subjects (neonate, 2yr, 5yr, 12yr) using established allometric equations (e.g., weight^0.75 for flows, weight^1.0 for volumes). 3. Adjust age-specific physiological factors: higher extracellular water fraction (neonates), lower plasma protein concentrations. 4. Incorporate the age-dependent CI-MPR expression factor as a modifier on the hepatic uptake rate constant (kint). 5. Run simulations administering the adult mg/kg dose to each virtual pediatric population. Compare PK exposure (AUC, Cmax). 6. Iteratively adjust the dose (mg/kg) in the pediatric models until exposures fall within ±20% of the adult target exposure. Propose weight-based dosing bands.
The Scientist's Toolkit: Key Research Reagent Solutions
- Validated Anti-CI-MPR (M6P Receptor) Antibody: For quantifying key clearance receptor expression across tissues and ages.
- Pediatric Physiological Parameter Database: Curated resource of age-stratified organ weights, blood flows, and composition data.
- Allometric Scaling Software/Tool: To systematically generate pediatric physiological parameters for PBPK platform.
- Banked Pediatric Tissue Biobank (Ethically Sourced): Critical for quantifying developmental changes in target biology.
Pediatric Dose Extrapolation via PBPK Workflow
Navigating Challenges: Common Pitfalls and Optimization Strategies in Biologics PBPK
Within the context of a broader thesis on Physiologically-Based Pharmacokinetic (PBPK) modeling for monoclonal antibodies (mAbs) and therapeutic proteins, managing model uncertainty and variability is paramount. These models are crucial for predicting human pharmacokinetics (PK), pharmacodynamics (PD), and first-in-human dose selection, but are subject to multiple sources of error and biological diversity. This application note details key sources and provides experimental protocols to quantify and reduce these uncertainties.
Physiological and System-Dependent Variability
This encompasses interspecies differences and inter-individual human variability in physiological parameters critical for mAb disposition (e.g., FcRn concentration, endothelial transcytosis rates, lymphatic flow, target antigen expression).
Drug-Specific Parameter Uncertainty
Uncertainty arises from in vitro measurements of critical parameters such as:
- Binding Affinity: Target antigen binding (KD), FcRn binding (pH-dependent).
- Non-Specific Interactions: Tissue uptake rate (Kp).
- Immunogenicity: Anti-drug antibody (ADA) incidence and impact.
Model Structure Uncertainty
This involves the selection of mathematical representations for key processes (e.g., linear vs. saturable target-mediated drug disposition (TMDD), intracellular trafficking mechanisms, interplay between lymphatic and vascular systems).
Table 1: Key Physiological Parameters Contributing to Inter-Individual Variability in mAb PK
| Parameter | Typical Value (Human) | Reported CV% | Primary Impact on PK |
|---|---|---|---|
| Plasma Volume | 40-55 mL/kg | ~15% | Initial Volume of Distribution (V1) |
| Lymph Flow Rate | 0.2-2.0 L/day | >50% | Subcutaneous Absorption, Distribution |
| FcRn Abundance (Endothelium) | 50-150 pmol/g tissue | ~40% | Half-life, Clearance |
| Target Antigen Density (Tissue) | Highly variable | 60-200% | Non-linear Clearance, Volume at Steady State (Vss) |
| Neonatal Fc Receptor (FcRn) Binding Affinity at pH 6.0 (KD) | 50-500 nM | ~30% | Endosomal Recycling, Half-life |
Table 2: Common Sources of Uncertainty in In Vitro to In Vivo Translation
| In Vitro Assay | Measured Parameter | Common Uncertainty Factor | Consequence for Model Prediction |
|---|---|---|---|
| Surface Plasmon Resonance (SPR) | Binding Kinetics (kon, koff, KD) | 2-5 fold | Inaccurate prediction of target occupancy & TMDD |
| Cell-based FcRn Recycling Assay | Fraction Recycled | 1.5-3 fold | Misestimation of half-life |
| In vitro Pinocytosis/Uptake Assay | Non-specific Endocytic Rate (Kp) | >10 fold | Uncertainty in tissue distribution & catabolic rate |
Experimental Protocols for Parameter Estimation and Uncertainty Reduction
Protocol: IntegratedIn Vitro–In SilicoEstimation of FcRn-Mediated Recycling Parameters
Objective: To accurately determine the pH-dependent binding affinity to human FcRn and the cellular recycling efficiency for a therapeutic mAb.
Materials: See "Scientist's Toolkit" (Section 7).
Workflow:
- SPR Binding Kinetics: Determine kon, koff, and KD at pH 6.0 and pH 7.4 using a Biacore system with immobilized recombinant human FcRn.
- Cell Recycling Assay: Use an engineered human endothelial cell line (e.g., HMEC-1) expressing human FcRn. Load cells with pH-sensitive fluorophore-labeled mAb at pH 6.0 for 30 min.
- Pulse-Chase & Flow Cytometry: Replace medium with pH 7.4 buffer to initiate recycling. Track remaining intracellular fluorescence via flow cytometry at time points (0, 2, 4, 8, 24 h).
- Data Fitting: Co-fit SPR and cellular recycling data to a mechanistic cellular recycling model within a PBPK framework using global fitting algorithms (e.g., Monte Carlo Parametric Expectation Maximization [MCPEM]) to estimate in vivo-relevant recycling rate constants and their confidence intervals.
Diagram Title: Integrated *In Vitro-In Silico FcRn Assay Workflow*
Protocol: Quantifying Target Antigen Expression and TurnoverIn Vivo
Objective: To characterize target antigen density and synthesis/degradation rates in relevant tissues to inform TMDD model structure and parameters.
Materials: See "Scientist's Toolkit" (Section 7).
Workflow:
- Tissue Sampling: Obtain relevant tissues (e.g., tumor, synovium) from animal models (humanized or disease-state) or human biopsies (where ethically feasible).
- Quantitative Immunofluorescence/ISH: Stain tissue sections with validated antibodies or probes against the target antigen and a cellular marker (e.g., CD31 for endothelium). Use digital pathology platforms for quantification.
- Flow Cytometry of Single-Cell Suspensions: Generate single-cell suspensions from tissues. Stain for target antigen and cell lineage markers. Use quantification beads to determine absolute antigen count per cell.
- Stable Isotope Labeling In Vivo: In animal models, administer heavy water (²H₂O) or labeled amino acids. Isolate target antigen via immunoprecipitation from tissues over a time course. Use mass spectrometry to measure incorporation rates and calculate synthesis and degradation rate constants (ksyn, kdeg).
- Model Integration: Integrate heterogeneous antigen density data and turnover rates into a whole-body PBPK-TMDD model, applying population variability distributions to key parameters.
Diagram Title: Workflow for Target Antigen Characterization In Vivo
Strategies for Structural Uncertainty and Model Qualification
Model Selection using SA/VSA:
Perform local Sensitivity Analysis (SA) and Global Variance-Based Sensitivity Analysis (VSA) to identify parameters dominating PK variability. Use Bayesian model selection criteria (e.g., Deviance Information Criterion - DIC) to compare nested and non-nested model structures (e.g., quasi-equilibrium vs. full TMDD).
Virtual Population Simulation:
Generate virtual patient populations by sampling from distributions of physiological and drug-specific parameters. Simulate expected PK variability and compare the envelope of simulations against observed population PK data from early clinical trials.
Diagram Title: Key mAb Pathways and Uncertainty Sources in PBPK
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for mAb PBPK Uncertainty Research
| Item | Function & Relevance to Uncertainty Reduction |
|---|---|
| Biacore 8K / Sartorius Gator | Gold-standard for label-free kinetic analysis of mAb-FcRn and mAb-target interactions. Reduces parameter uncertainty. |
| Recombinant Human FcRn | Critical reagent for in vitro binding assays. Batch-to-batch consistency is key for reproducible KD measurements. |
| Engineered FcRn-Expressing Cell Lines (e.g., HMEC-1-hFcRn) | Provides a cellular context for recycling assays, bridging in vitro binding and in vivo disposition. |
| Quantitative Immunofluorescence Kits (e.g., Akoya/Ultivue) | Enable multiplex, absolute quantification of target antigen density and distribution in tissue, informing spatial model parameters. |
| Calibrated Quantification Beads for Flow Cytometry (e.g., Quantum MESF beads) | Allow conversion of fluorescence intensity to absolute antigen counts per cell, reducing inter-assay variability. |
| Stable Isotope Labels (²H₂O, ¹³C-Lysine) | Used in vivo to measure target antigen synthesis and degradation rates (ksyn, kdeg), critical for TMDD models. |
| Monolith Nano / MicroScale Thermophoresis (MST) | Alternative for measuring binding affinities in solution, useful for difficult-to-immobilize targets. |
| Software: MATLAB/Simbiology, R/mrgsolve, Certara Phoenix | Platforms for implementing complex PBPK models, performing SA/VSA, and population PK/PD analysis. |
Handling Parameter Identifiability Issues with Complex TMDD Systems
1. Introduction within a PBPK Thesis Context Within the broader thesis on developing comprehensive Physiologically-Based Pharmacokinetic (PBPK) models for monoclonal antibodies (mAbs) and therapeutic proteins, addressing the Target-Mediated Drug Disposition (TMDD) mechanism is critical. TMDD models describe nonlinear PK arising from high-affinity target binding. However, their full mathematical representations are often over-parameterized, leading to identifiability issues where unique parameter estimation from available data is impossible. This application note provides protocols to diagnose and resolve these issues, ensuring robust model development for translational research.
2. Key Quantitative Data on TMDD Model Structures
Table 1: Comparison of TMDD Model Simplifications and Their Impact on Identifiability
| Model | Key Parameters | Required Data for Identifiability | Common Identifiability Issue | Typical Use Case |
|---|---|---|---|---|
| Full TMDD | kon, koff, kint, Rtot, kdeg | Rich data: free drug, total drug, total target (bound+free) | kon and Rtot often correlated; koff and kint correlated. | Early research with abundant biomarker data. |
| Michaelis-Menten (MM) | Vmax, KM | Only total drug concentration PK data. | Structurally identifiable but may mask underlying biology. | Late-stage development, clinical PK analysis. |
| Quasi-Equilibrium (QE) | KD, kint, Rtot | Total drug concentration, assumes rapid binding. | KD and Rtot may be correlated if kint ~ kdeg. | When binding is fast relative to other processes. |
| Quasi-Steady State (QSS) | KSS, kint, Rtot | Total drug concentration, assumes slow internalization. | Improved over QE but correlation persists with sparse data. | Standard for mAbs with observable linear phase. |
Table 2: Common Diagnostics for Assessing Parameter Identifiability
| Diagnostic Method | Measurement/Output | Interpretation | Threshold/Significance |
|---|---|---|---|
| Correlation Matrix | Parameter correlation coefficient (r) | Absolute r > 0.9 suggests strong collinearity and potential non-identifiability. | |
| Coefficient of Variation (CV%) | CV% from covariance matrix estimation. | CV% > 50% indicates poor practical identifiability for that parameter. | |
| Profile Likelihood | Log-likelihood vs. fixed parameter value. | A flat profile indicates structural non-identifiability. A shallow, but unique minimum suggests poor practical identifiability. | |
| Fisher Information Matrix (FIM) | Rank of FIM / Eigenvalues. | Rank deficiency vs. parameter count = structural non-identifiability. Small eigenvalues = poor practical identifiability. |
3. Experimental Protocols
Protocol 1: Structural Identifiability Analysis Using the Profiling Method Objective: To determine if model parameters are uniquely identifiable from the proposed experimental design. Materials: TMDD model code (e.g., in NONMEM, Monolix, or R), dataset (real or simulated design). Procedure:
- Model Fitting: Fit the full TMDD model to the data to obtain the maximum likelihood estimate (MLE) for all parameters.
- Parameter Fixing: Select one parameter of interest (θi). Fix it at a range of values around its MLE (e.g., ± 50%).
- Re-optimization: For each fixed value of θi, re-optimize all other free model parameters to minimize the objective function value (OFV).
- Profile Calculation: Record the resulting OFV for each value of θi. Calculate the profile log-likelihood: PL(θi) = -0.5 * (OFV(θi) - OFVMLE).
- Plot & Assess: Plot PL(θi) against θi. A uniquely identifiable parameter will show a sharp, V-shaped minimum. A flat profile indicates non-identifiability.
- Iterate: Repeat steps 2-5 for all key model parameters.
- Conclusion: Parameters with flat profiles must be fixed, simplified, or their estimation requires additional data types.
Protocol 2: Practical Identifiability Assessment via Bootstrap Analysis Objective: To evaluate the precision and potential correlations of parameter estimates given the noise and sparsity of typical data. Materials: Finalized model, original estimation dataset, statistical software with bootstrapping capabilities. Procedure:
- Dataset Resampling: Generate 500-1000 bootstrap replicates by randomly sampling subjects from the original dataset with replacement.
- Model Re-estimation: Fit the TMDD model (potentially a simplified, identifiable version) to each bootstrap sample.
- Parameter Distribution: Collect the estimated parameter values from each successful run.
- Diagnostic Calculation:
- Calculate the median and 95% confidence interval (2.5th to 97.5th percentile) for each parameter.
- Compute the empirical correlation matrix from the bootstrap estimates.
- Interpretation: Parameters with wide confidence intervals (e.g., spanning an order of magnitude) are practically non-identifiable. High correlation magnitudes (>0.9) between parameters confirm collinearity issues.
4. Visualizations
Diagram 1: Decision workflow for handling TMDD identifiability
Diagram 2: Full TMDD system pathways and parameters
5. The Scientist's Toolkit
Table 3: Key Research Reagent Solutions for TMDD Model Validation
| Item | Function in TMDD Research | Application Note |
|---|---|---|
| SPR/Biacore System | Measures binding kinetics (kon, koff, KD) in vitro. | Provides prior information to fix highly correlated parameters (e.g., koff), resolving structural non-identifiability. |
| Quantitative Target ELISA/Ligand Binding Assay | Measures free and/or total target concentration in serum/tissue. | Enriches PK data, enabling estimation of Rtot and kdeg separately, breaking parameter correlations. |
| Anti-idiotype mAb Assay | Specifically measures free drug concentration in the presence of target. | Allows direct verification of model-predicted free drug profiles, strengthening model credibility. |
| Stable, Labeled Protein Standards (SEAP, Fc-fusion reporters) | Acts as a target surrogate to monitor cellular internalization and recycling (kint, krecycle). | Informs system-specific parameters in cell-based systems, reducing uncertainty. |
| PBPK Software with TMDD Module (e.g., PK-Sim, Simbiology, GastroPlus) | Integrates TMDD into whole-body physiology. | Facilitates translation from rich pre-clinical data (with identifiability) to sparser clinical data via system-informed priors. |
1.0 Introduction & Thesis Context This document provides application notes and protocols for optimizing physiologically-based pharmacokinetic (PBPK) model performance, specifically within a broader thesis on PBPK modeling for monoclonal antibodies (mAbs) and therapeutic proteins. Robust parameter estimation and sensitivity analysis (SA) are critical for developing credible, predictive models that inform drug development decisions, from preclinical candidate screening to clinical dose regimen design.
2.0 Key Parameter Estimation Strategies Successful PBPK model development for large molecules requires the systematic identification of key physiological and drug-specific parameters. Primary estimation methods are summarized below.
Table 1: Core Parameter Estimation Methods for mAb PBPK Models
| Parameter Category | Example Parameters | Primary Estimation Method | Typical Data Source |
|---|---|---|---|
| Physiological | Vascular/Interstitial volumes, lymph flow rates, FcRn expression | In silico system priors | Population physiology literature, proteomics data |
| Drug-Specific | Target binding affinity (Kd), internalization rate | In vitro assays | Surface Plasmon Resonance (SPR), cell-based assays |
| Systemic PK | Linear clearance, FcRn affinity (Kd_FcRn), endocytic rate | In vivo fitting & pooling | Preclinical PK in relevant animal models (e.g., humanized FcRn mice, non-human primates) |
| Tissue-Specific | Permeability-surface area product, target expression | Hybrid in vitro-in vivo | Biodistribution studies, quantitative tissue imaging |
2.1 Protocol: In Vivo Pooled Parameter Estimation from Preclinical PK
- Objective: Estimate systemic clearance and steady-state volume of distribution (Vss) parameters by simultaneously fitting multi-dose PK data.
- Materials: PK serum concentration-time data from at least three dose levels (e.g., 1, 10, 50 mg/kg) in relevant animal model(s).
- Procedure:
- Data Compilation: Pool all serum concentration-time data across doses and animals into a single dataset.
- Model Definition: Use a minimal PBPK model structure (e.g., two-compartment model with linear and nonlinear FcRn salvage).
- Objective Function: Define a log-likelihood or weighted least squares objective function. Apply appropriate weighting (e.g., 1/y_obs²) for heteroscedastic data.
- Algorithm Selection: Employ a global optimization algorithm (e.g., Particle Swarm, Genetic Algorithm) to explore parameter space widely, followed by a local method (e.g., Nelder-Mead) for refinement.
- Diagnostic Checks: Assess goodness-of-fit via visual predictive checks, residual plots, and parameter correlation matrices.
3.0 Sensitivity Analysis Best Practices SA quantifies how uncertainty in model inputs (parameters) propagates to uncertainty in model outputs (e.g., AUC, Cmax, tissue exposure).
Table 2: Comparison of Sensitivity Analysis Techniques
| Method | Description | Use Case | Computational Cost |
|---|---|---|---|
| Local SA (One-at-a-Time) | Varies one parameter at a time around a nominal value. | Quick screening of influential parameters; Jacobian matrix for estimation. | Low |
| Global SA (e.g., Sobol', Morris) | Varies all parameters simultaneously over their full distributions. | Ranking key uncertainty sources; identifying interactions. | High (requires thousands of runs) |
| Extended Fourier Amplitude (eFAST) | Spectral analysis method to compute total-order sensitivity indices. | Reliable main & total effect indices for nonlinear models. | Medium-High |
3.1 Protocol: Global Sensitivity Analysis Using the Morris Screening Method
- Objective: Rank parameters by their influence on key model outputs to guide future research and refinement efforts.
- Materials: A qualified PBPK model, defined parameter ranges (min, max), and a chosen model output (e.g., tumor AUC).
- Procedure:
- Parameter Selection & Ranges: Select all uncertain parameters (e.g., 10-20). Define a physiologically plausible range for each (e.g., ± 30% of nominal).
- Trajectory Generation: Generate
rtrajectories (typically 50-100) in parameter space using the Elementary Effects method. Each trajectory involvesp+1model simulations, wherepis the number of parameters. - Model Execution: Run the PBPK model for each parameter set defined by the trajectories.
- Elementary Effect Calculation: For each parameter
iin trajectoryk, compute:EE_i^k = [Y(P1,...,Pi+Δ,...,Pp) - Y(P)] / Δ, whereΔis a perturbation factor. - Sensitivity Metrics: Calculate the mean (
μ*) of the absolute Elementary Effects (measuring overall influence) and the standard deviation (σ) of the Elementary Effects (measuring interaction/nonlinearity). Rank parameters byμ*.
4.0 The Scientist's Toolkit: Research Reagent Solutions Table 3: Essential Materials for mAb PBPK Model Development & Validation
| Item | Function in PBPK Context |
|---|---|
| Human FcRn Transgenic Mouse Model | In vivo system for estimating human-FcRn dependent PK parameters (clearance, half-life). |
| Biolayer Interferometry (BLI) or SPR System | Label-free in vitro assay to quantify critical binding kinetics (e.g., mAb:FcRn, mAb:Target). |
| Quantitative Whole-Body Autoradiography (QWBA) | Provides spatial, quantitative tissue distribution data for model validation, especially for non-blood compartments. |
| LC-MS/MS with Stable Isotope Labeled mAb Internal Standard | Gold-standard for precise, specific quantification of therapeutic protein concentrations in complex biological matrices. |
| Population-Based PBPK Software (e.g., PK-Sim, Simbiology) | Platform for implementing full PBPK models, performing parameter estimation, and running virtual population simulations. |
5.0 Visualized Workflows
PBPK Model Development and Optimization Cycle
mAb Tissue Disposition and FcRn Salvage Pathways
This application note, framed within a thesis on PBPK modeling for monoclonal antibodies (mAbs) and therapeutic proteins, details strategies to address critical data gaps in preclinical development. The transition from in vitro assays and in silico predictions to accurate in vivo extrapolation remains a key challenge. Here, we outline integrated protocols and reagent solutions to generate high-quality input data for PBPK models, enhancing the prediction of human pharmacokinetics and pharmacodynamics.
Core Quantitative Data for PBPK Inputs
The following tables summarize essential quantitative parameters required for mAb PBPK modeling, often derived from in vitro experiments.
Table 1: Key In Vitro Biophysical and Binding Assay Parameters
| Parameter | Typical Assay | Relevance to PBPK Model | Common Value Range (mAbs) |
|---|---|---|---|
| Target Affinity (KD) | Surface Plasmon Resonance (SPR) | Determines binding kinetics for target-mediated drug disposition (TMDD). | 0.01 - 10 nM |
| Association Rate (kon) | SPR, Bio-Layer Interferometry (BLI) | Input for kinetic TMDD models. | 1e4 - 1e6 M⁻¹s⁻¹ |
| Dissociation Rate (koff) | SPR, BLI | Determines complex stability. | 1e-5 - 1e-3 s⁻¹ |
| FcRn Affinity (pH 6.0) | SPR | Predicts FcRn-mediated recycling and half-life. | KD: 100-1000 nM |
| Non-specific Binding | ELISA, MSD | Informs tissue partitioning coefficients. | Varies by assay |
| Isoelectric Point (pI) | Imaged Capillary IEF | Predicts electrostatic tissue interactions. | 7.0 - 9.5 |
Table 2: Critical In Vitro Cellular and Transporter Assay Parameters
| Parameter | Assay System | PBPK Model Input | Notes |
|---|---|---|---|
| Cell-based Internalization Rate | Live-cell imaging, flow cytometry | Cellular uptake rate constant (kint). | Use antigen-positive cell lines. |
| Lysosomal Degradation Rate | Pulse-chase, catabolism assays | Degradation rate constant (kdeg). | Often coupled with internalization. |
| FcRn-mediated Recycling Efficiency | pH-switch assays, transcytosis models | Fraction recycled vs. degraded. | Key for half-life prediction. |
| Off-target Binding/ Uptake | Primary cell co-cultures, tissue sections | Non-specific clearance component. | Assess via flow cytometry or imaging. |
Detailed Experimental Protocols
Protocol 3.1: Determining Cell-Based Internalization and Degradation Kinetics
Objective: To quantify the rate of antibody internalization and subsequent lysosomal degradation in target-expressing cells for estimating kint and kdeg.
Materials: See "Scientist's Toolkit" (Section 5). Procedure:
- Cell Preparation: Seed target-positive cells (e.g., HER2+ SK-BR-3 for trastuzumab studies) in 12-well plates at 2.5 x 10^5 cells/well. Culture overnight to achieve 80% confluence.
- Surface Labeling (Pulse): Chill cells on ice. Wash twice with ice-cold FACS buffer. Incubate with 10 µg/mL fluorescently labeled mAb (e.g., Alexa Fluor 647 conjugate) in buffer for 1 hour on ice. Perform triplicate wells for each time point.
- Initiate Internalization (Chase): Remove unbound antibody with three ice-cold washes. Add pre-warmed complete media and immediately transfer plates to a 37°C, 5% CO2 incubator to initiate internalization.
- Time-course Sampling: At defined time points (e.g., 0, 15, 30, 60, 120, 240 min), terminate internalization by placing the well on ice.
- Acid-Strip Surface Antibody: Wash cells with ice-cold PBS. Treat with acid-strip buffer (0.5 M NaCl, 0.2 M acetic acid, pH 2.5) for 5 minutes on ice to remove remaining surface-bound antibody. Neutralize with complete media.
- Flow Cytometry Analysis: Harvest cells using gentle detachment. Analyze median fluorescence intensity (MFI) of the internalized fraction using a flow cytometer.
- Data Fitting: Plot internalized MFI vs. time. Fit data to a first-order kinetics model: Internalized Signal = A(1 - e^{-kintt}), where *kint is the internalization rate constant.
- Degradation Assessment (Extended Chase): For degradation, extend chase times to 24-48 hours, measuring residual intracellular fluorescence. Fit decay phase to estimate degradation rate constant (kdeg).
Protocol 3.2: FcRn Affinity and Recycling Assay Using Surface Plasmon Resonance (SPR)
Objective: To measure the pH-dependent binding affinity of a mAb to human FcRn, a critical parameter for predicting serum half-life.
Materials: Biacore or equivalent SPR system, CMS chip, human FcRn protein, running buffers (pH 6.0: 50 mM MES, 50 mM NaCl; pH 7.4: PBS). Procedure:
- Chip Functionalization: Immobilize anti-His antibody (~10,000 RU) on a CMS chip using standard amine coupling to capture His-tagged FcRn.
- Capture Cycle: Inject His-tagged human FcRn (5 µg/mL) for 60 seconds at pH 6.0 running buffer to achieve a consistent capture level (~100 RU).
- Analyte Binding: Inject a concentration series of the mAb (0.78 nM to 200 nM) over the FcRn surface for 120 seconds at pH 6.0.
- Dissociation & Regeneration: Monitor dissociation in pH 6.0 buffer for 300 seconds. Regenerate the surface with a 30-second injection of pH 7.4 buffer to dissociate the mAb-FcRn complex, followed by re-equilibration at pH 6.0.
- Reference Subtraction: Perform identical injections over a reference flow cell without captured FcRn. Subtract reference sensorgrams.
- Kinetic Analysis: Fit the subtracted sensorgrams globally to a 1:1 Langmuir binding model using the SPR evaluation software to derive kon, koff, and KD at pH 6.0.
- Specificity Confirmation: Run a control injection at pH 7.4 to confirm negligible binding, characteristic of FcRn salvage pathway specificity.
Visualization of Pathways and Workflows
Title: Integrated IVIVE and PBPK Modeling Workflow
Title: Key Cellular Pathways Governing mAb PK
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Key Protocols
| Item/Reagent | Function in IVIVE for PBPK | Example Product/Source |
|---|---|---|
| Biacore T200 / Nicoya Lifesci OpenSPR | Measures real-time biomolecular interactions (kinetics, affinity) for TMDD & FcRn binding. | Cytiva Biacore T200; Nicoya OpenSPR. |
| His-tagged Human FcRn (heterodimer) | Critical reagent for quantifying pH-dependent binding affinity in SPR assays. | Sino Biological, 10782-H08H; Themo Fisher, RM3257. |
| pH-Switch Assay Buffer Kit | Provides buffers at precise pH (6.0 vs 7.4) to mimic endosomal and physiological conditions. | FabGennix IFD-001. |
| Fluorescent Antibody Labeling Kit (Alexa Fluor 647) | Labels mAbs for visualization and quantification in cellular uptake/degradation assays. | Thermo Fisher, A20186. |
| Target-positive Cell Line (e.g., SK-BR-3) | Cellular model expressing relevant antigen for measuring internalization kinetics (kint). | ATCC, HTB-30. |
| Imaged Capillary Isoelectric Focusing (icIEF) System | Precisely determines pI, influencing charge-based tissue distribution in PBPK models. | ProteinSimple, Maurice. |
| Primary Human Endothelial Cell Systems | Assess non-specific uptake and transcytosis in physiologically relevant barriers. | PromoCell, C-12210. |
| PBPK Software Platform (with mAb capability) | Integrates in vitro parameters to perform IVIVE and simulate human PK. | Simcyp Simulator, GastroPlus. |
Within the broader thesis on advancing PBPK modeling for monoclonal antibodies (mAbs) and therapeutic proteins, this document details the critical process of model refinement. The transition from preclinical (animal) data to clinical (human) data is not linear but iterative. Each phase of drug development generates new data that must be systematically integrated to refine and validate the PBPK model, enhancing its predictive power for pharmacokinetics (PK), pharmacodynamics (PD), and safety in patients.
Core Principles of the Iterative Refinement Cycle
The refinement cycle is built on four pillars: Predict, Compare, Analyze, and Update.
- Predict: Use the current model version to simulate outcomes for the next experimental or clinical phase.
- Compare: Systematically compare model predictions with newly observed in vivo data.
- Analyze: Quantify discrepancies, identify bias, and hypothesize mechanistic gaps (e.g., unsaturating target-mediated drug disposition, immune responses, disease effects).
- Update: Refine model structure and parameters (e.g., FcRn affinity, endocytic rate, target expression) to reconcile predictions with data. This updated model becomes the basis for the next cycle.
Data Integration and Quantitative Comparison Tables
Table 1: Preclinical to Clinical Data Streams for PBPK Model Refinement
| Data Tier | Source System | Key PK Parameters Inferred | Key PD/Safety Endpoints | Primary Role in Refinement |
|---|---|---|---|---|
| Preclinical In Vitro | Target binding assays, cell lines (e.g., HEK293), human tissue lysates | Target affinity (KD), internalization rate (kint) | Cell proliferation/ inhibition | Initialize Model: Provide system-independent parameters. |
| Preclinical In Vivo | Mouse, rat, cynomolgus monkey PK studies; disease models | Clearance (CL), Volume (Vss), nonlinear PK parameters | Efficacy (e.g., tumor size), target engagement, ADA incidence | Translate & Calibrate: Scale parameters, identify species-specific pathways for humanization. |
| Phase I Clinical | First-in-Human SAD/MAD trials in healthy volunteers or patients | Human CL, Vss, linear/nonlinear PK profile, half-life (t1/2) | Safety, tolerability, immunogenicity (ADA) | Ground-Truth Core PK: Calibrate system-specific human physiology (e.g., plasma volume, FcRn concentration). |
| Phase II/III Clinical | Patient populations, various dosing regimens | Population variability (BSV), covariate effects (weight, albumin, TMDD), drug-drug interactions | Clinical efficacy, safety signals, immunogenicity impact | Validate & Extrapolate: Refine for disease physiology, predict optimal dosing for subgroups. |
Table 2: Example Discrepancy Analysis & Model Update Actions
| Observed vs. Predicted Discrepancy | Potential Mechanistic Gap | Model Refinement Action |
|---|---|---|
| Human clearance is 2x faster than predicted from monkey allometric scaling. | Unsaturated non-specific pinocytosis or higher endothelial uptake in humans. | Increase first-order pinocytotic rate constant (kpin) in human tissue compartments. |
| Terminal half-life decreases at higher dose levels in clinic. | Target-mediated drug disposition (TMDD) not fully captured in preclinical low-dose studies. | Implement full TMDD model with accurate estimate of total target pool size in human tissues. |
| Higher exposure variability in patient population vs. healthy volunteers. | Disease state (e.g., inflammation) altering FcRn recycling or vascular permeability. | Introduce disease-specific scaling factor on FcRn expression or lymph flow rate. |
| Late-onset anti-drug antibodies (ADA) reducing exposure in later cycles. | ADA-enhanced clearance not included in model. | Add an ADA-driven clearance pathway, triggered after a time-dependent immune response. |
Detailed Experimental Protocols
Protocol 4.1:In VivoPK Study in Non-Human Primates for mAb Clearance Estimation
Objective: To obtain preclinical PK data for human clearance prediction and model calibration. Materials: See Scientist's Toolkit. Procedure:
- Animal Preparation: Use cynomolgus monkeys (n=3-4 per dose group). House under standard conditions. Insert vascular access ports (VAPs) for serial blood sampling.
- Dosing: Administer the therapeutic mAb intravenously at three dose levels (e.g., 1, 10, 50 mg/kg) to assess linearity.
- Sample Collection: Collect blood samples pre-dose and at 0.083, 0.25, 0.5, 1, 2, 4, 8, 24, 48, 72, 96, 168, 240, and 336 hours post-dose.
- Sample Processing: Centrifuge blood at 1500×g for 10 min at 4°C. Aliquot serum and store at -80°C.
- Bioanalysis: Quantify mAb serum concentrations using a validated ligand-binding assay (e.g., ELISA or MSD).
- Non-Compartmental Analysis (NCA): Calculate AUC0-∞, CL, Vss, and t1/2.
- Model Fitting: Fit the PBPK model to concentration-time data using a nonlinear mixed-effects approach (e.g., Monolix, NONMEM) to estimate parameters like CLlinear, CLtarget, and koff,FcRn.
Protocol 4.2: Clinical First-in-Human Study for Model Grounding
Objective: To obtain definitive human PK parameters and refine the PBPK model. Design: Randomized, placebo-controlled, single ascending dose (SAD) study. Procedure:
- Cohorts: Enroll 8 healthy volunteers per cohort (6 active:2 placebo). Start dose at 1/50th of NOAEL from NHP studies.
- Dosing & Sampling: Administer single IV dose. Collect intensive PK samples over 8-12 weeks (to capture 3-5 half-lives).
- Assay: Use a validated, GLP-compliant pharmacokinetic assay for human serum.
- Population PK (PopPK) Analysis: Develop a base PopPK model (typically 2-compartment with linear/nonlinear clearance).
- PBPK Model Refinement: Use the observed human concentration-time profiles as the optimization target. Adjust system-specific human parameters within physiological bounds (e.g., lymphatic flow rates, tissue volumes) and refine drug-specific parameters (e.g., endosomal degradation rate) until predictions align with observed data. Validate using visual predictive checks.
Visualizations
Diagram 1: Iterative PBPK Model Refinement Cycle Across Drug Development
Diagram 2: Data Integration into the PBPK Model Core for Prediction
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for PBPK Refinement Studies
| Item | Function in Model Refinement | Example Product/Source |
|---|---|---|
| Recombinant Human Target Protein | Used in in vitro assays (SPR, ELISA) to measure binding affinity (KD), a critical drug-specific parameter. | Sino Biological, R&D Systems. |
| Anti-Drug Antibody (ADA) Assay Kit | To detect and quantify immunogenicity in preclinical and clinical serum samples, informing ADA clearance models. | Meso Scale Discovery (MSD) Bridging Assay Kit. |
| Species-Specific IgG/Anti-IgG ELISA Kits | For accurate quantification of mAb concentrations in serum/plasma from different animal species during PK studies. | AlphaLISA (PerkinElmer), Species-Specific IgG Quantitation Kits. |
| PBPK Modeling Software | Platform for building, simulating, and fitting PBPK models to iterative data sets. | GastroPlus (Simulations Plus), PK-Sim (Open Systems Pharmacology), Simbiology (MATLAB). |
| Population PK/PD Analysis Software | For statistical analysis of clinical trial data, estimation of population parameters & variability, used to inform PBPK. | NONMEM, Monolix, Phoenix NLME. |
| Cynomolgus Monkey FcRn Affinity Column | To experimentally determine the FcRn binding affinity of the mAb in a relevant species, refining FcRn recycling parameters. | Capturem FcRn Affinity Resin (Takara Bio). |
| Human Tissue Homogenates | Used to assess non-specific tissue binding and distribution coefficients (Kp) for the mAb. | XenoTech, BioIVT. |
Ensuring Confidence: Validating PBPK Models and Comparing Modeling Paradigms
This application note details a comprehensive validation framework for PBPK models of monoclonal antibodies (mAbs) and therapeutic proteins, critical for regulatory acceptance and reliable prediction of human pharmacokinetics (PK).
Validation Framework: Core Definitions and Metrics
A robust validation strategy progresses through three sequential tiers.
Table 1: Tiers of PBPK Model Validation for mAbs/Therapeutic Proteins
| Validation Tier | Definition & Purpose | Key Quantitative Acceptance Criteria |
|---|---|---|
| Internal | Verification that the model can accurately describe the data used to build it (e.g., single-species PK). Ensures mathematical and coding integrity. | Visual predictive check (VPC): ≥90% of observed data points within 90% prediction interval. Objective function value (OFV) minimization. |
| External | Evaluation of model performance against a distinct dataset not used in model development (e.g., PK from a different study or population). Tests predictive power. | Prediction error (PE): ≤2-fold for PK parameters (AUC, Cmax). Mean absolute percentage error (MAPE) < 30-40%. |
| Prospective | Prediction of clinical outcomes in a new scenario (e.g., first-in-human PK, special populations, drug-drug interactions) prior to data collection. | Clinical study results fall within the model's simulated prediction intervals, validating translational utility. |
Experimental Protocols for Key Supporting Assays
Accurate model parameters are derived from in vitro and in vivo experiments.
Protocol 2.1: In Vitro Binding Affinity (KD) Determination via Surface Plasmon Resonance (SPR)
- Objective: To quantify the binding affinity of the mAb to its soluble target antigen.
- Materials: Biacore SPR system, CMS sensor chip, target antigen, mAb, HBS-EP+ running buffer.
- Procedure:
- Activate the sensor chip surface with EDC/NHS.
- Immobilize the target antigen onto one flow cell via amine coupling; use another as a reference.
- Dilute the mAb to a series of concentrations (e.g., 0.78 nM to 100 nM) in running buffer.
- Inject mAb samples over the antigen and reference surfaces at a flow rate of 30 µL/min.
- Monitor the association phase (180 s), followed by dissociation in running buffer (300 s).
- Regenerate the surface with 10 mM Glycine-HCl, pH 1.5.
- Fit the resulting sensograms to a 1:1 Langmuir binding model to calculate the association (ka) and dissociation (kd) rate constants. KD = kd/ka.
Protocol 2.2: In Vivo Neonatal Fc Receptor (FcRn) Affinity Assessment in Transgenic Mice
- Objective: To assess the role of FcRn-mediated recycling on mAb half-life in vivo.
- Materials: Human FcRn transgenic mouse model, wild-type control mice, test mAb, ELISA reagents.
- Procedure:
- Administer a single intravenous dose (e.g., 10 mg/kg) of the test mAb to human FcRn transgenic mice (n=5) and wild-type controls (n=5).
- Collect serial blood samples at pre-dose, 0.083, 1, 24, 72, 168, 240, and 336 hours post-dose.
- Process samples to obtain plasma.
- Quantify mAb concentrations in all samples using a validated target-capture ELISA.
- Perform non-compartmental analysis (NCA) to estimate terminal half-life (t1/2) and AUC.
- Compare t1/2 and clearance between transgenic and wild-type groups. A significantly longer t1/2 in FcRn transgenic mice confirms functional FcRn interaction.
Validation Workflow and Pathway Diagrams
Title: Three-Tier PBPK Model Validation Sequential Workflow
Title: Key Pathways Governing mAb PK in a PBPK Model
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for mAb PBPK Model Parameterization
| Reagent / Material | Function in PBPK Context |
|---|---|
| Recombinant Human Target Antigen | Critical for in vitro assays (SPR, ELISA) to determine binding kinetics (KD, kon, koff) for TMDD model component. |
| Anti-Idiotypic Capture Antibodies | Enable development of specific PK ELISAs for quantifying mAb concentrations in complex biological matrices. |
| Human FcRn Transgenic Mouse Model | In vivo system to empirically assess the impact of FcRn affinity on mAb clearance and half-life for model refinement. |
| Physiologically Relevant Buffer Kits (pH 6.0 & 7.4) | For in vitro FcRn binding assays, simulating endosomal and physiological pH conditions. |
| Validated PBPK Software Platform (e.g., GastroPlus, Simcyp, PK-Sim) | Provides the structural framework, system parameters, and algorithms to integrate experimental data into a predictive model. |
Defining Acceptance Criteria and Performance Metrics for Large Molecule Models
Within the broader thesis on advancing Physiologically-Based Pharmacokinetic (PBPK) modeling for monoclonal antibodies (mAbs) and therapeutic proteins, a critical step is the rigorous definition of model acceptance criteria and performance metrics. This framework ensures that developed models are reliable, predictive, and suitable for supporting critical decisions in drug development, from lead optimization to clinical dose selection. This document provides application notes and detailed protocols for establishing these quantitative benchmarks, focusing on large molecule-specific challenges such as target-mediated drug disposition (TMDD), FcRn recycling, and immunogenicity.
Core Acceptance Criteria and Performance Metrics
The following table summarizes key quantitative metrics and their proposed acceptance criteria for validating large molecule PBPK models. These benchmarks are synthesized from current industry white papers, regulatory guidelines, and scientific literature.
Table 1: Performance Metrics and Acceptance Criteria for Large Molecule PBPK Models
| Metric | Formula/Description | Acceptance Criterion | Rationale for Large Molecules |
|---|---|---|---|
| Average Fold Error (AFE) | ( AFE = 10^{\frac{1}{n} \sum \log_{10}(\frac{Predicted}{Observed})} ) | 0.8 – 1.25 | Measures geometric mean bias. Critical for AUC predictions. |
| Absolute Average Fold Error (AAFE) | ( AAFE = 10^{\frac{1}{n} \sum \lvert \log_{10}(\frac{Predicted}{Observed}) \rvert} ) | ≤ 1.5 – 2.0* | Measures precision. A wider threshold (e.g., 2.0) may be accepted for early-phase TMDD models. |
| Root Mean Square Error (RMSE) | ( RMSE = \sqrt{\frac{\sum{i=1}^{n}(Predictedi - Observed_i)^2}{n}} ) | Context-dependent; ≤ 20% of mean observed value | Absolute measure of error magnitude. Useful for simulation-based validation. |
| Visual Predictive Check (VPC) | Overlay of observed percentiles (e.g., 5th, 50th, 95th) with model-simulated prediction intervals. | ≥ 90% of observed data points within the 90% prediction interval | Gold standard for population models. Assesses model capture of central trend and variability. |
| R-squared (R²) | Coefficient of determination for observed vs. predicted plots. | > 0.75 – 0.90 | Indicates proportion of variance explained. Can be misleading for non-linear systems. |
| Precision of Parameter Estimates | Relative Standard Error (RSE%) from model estimation. | RSE% < 30% for structural parameters; < 50% for variance parameters | Ensures parameter identifiability, crucial for complex TMDD models. |
*Note: Acceptance criteria may be phased, with stricter limits (e.g., AAFE ≤ 1.5) for final validation of models intended for regulatory submission.
Experimental Protocols for Model Verification
Protocol 2.1: Conducting a Visual Predictive Check (VPC) for a Population mAb PBPK Model
Objective: To validate that a developed population PBPK model adequately captures both the central tendency and the variability of observed pharmacokinetic data.
Materials & Software:
- Population PBPK model (e.g., in platforms like GastroPlus, Simcyp Simulator, or PK-Sim).
- Observed clinical PK dataset (e.g., from a Phase 1 study).
- Software for simulation and visualization (R, Python, or built-in simulator tools).
Procedure:
- Finalize Model: Fix all population model parameters (system, drug, variability).
- Design Simulation: Replicate the design of the clinical study (doses, regimens, subject demographics, sample times) exactly.
- Generate Simulations: Perform N=1000 trial replications using the same number of virtual subjects as the original study.
- Calculate Percentiles: For each time point, calculate the 5th, 50th (median), and 95th percentiles of the simulated concentration profiles across all trials.
- Construct Prediction Intervals: The area between the 5th and 95th simulated percentiles forms the 90% prediction interval.
- Overlay Observed Data: Plot the observed clinical data (often as percentiles or individual points) on the same graph.
- Assessment: Qualitatively assess if the observed data percentiles (e.g., median and spread) fall within the model's simulated prediction intervals. Quantitatively, calculate the percentage of observed data points lying inside the 90% prediction interval.
Protocol 2.2: Assessing Predictive Performance via External Validation
Objective: To test model predictive power using a dataset not used for model calibration (e.g., a different patient population or dosing regimen).
Procedure:
- Dataset Selection: Secure a robust external PK dataset (e.g., a different dose, patient population with renal impairment, or pediatric data).
- Blind Prediction: Without re-estimating any parameters, simulate the PK profile for the external study design.
- Calculate Metrics: Compute AFE, AAFE, and RMSE (Table 1) comparing predictions to the new observed data.
- Apply Acceptance Criteria: Evaluate if the metrics meet pre-defined acceptance criteria. Failure indicates limited model generalizability and may require structural refinement.
Visualization of Key Concepts
Diagram 1: Validation Workflow for mAb PBPK Model
Diagram 2: Key Processes in mAb Disposition for Model Building
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Tools for Large Molecule PBPK Model Development & Validation
| Item | Function in Context |
|---|---|
| Specialized PBPK Software (e.g., Simcyp, GastroPlus, PK-Sim) | Provides built-in, validated systems biology platforms with pre-defined large molecule (mAb) modules incorporating FcRn recycling, lymph flow, and TMDD models. |
| In Vitro Assay Kits (FcRn Binding Affinity, Target Binding Kinetics) | Generates critical input parameters (e.g., KD, Kon/Koff) for model parameterization, reducing reliance on in vivo fitting. |
| Anti-Idiotypic Antibodies | Essential reagents for quantifying free vs. total therapeutic antibody concentrations in complex PK/PD assays, informing model structure. |
| Recombinant Human Targets & FcRn Proteins | Used in surface plasmon resonance (SPR) or kinetics exclusion assays to measure binding constants in vitro for model input. |
| Population Database with IgG/ADA Levels | Integrated demographic databases (e.g., within PBPK platforms) allow simulation of covariates like baseline endogenous IgG or ADA incidence on mAb PK. |
| Clinical PK/PD Datasets (Public/Internal) | Serve as the gold standard for model calibration (internal) and rigorous external validation to establish credibility. |
Within the broader thesis on PBPK modeling for monoclonal antibodies (mAbs) and therapeutic proteins, a critical methodological decision lies in selecting the appropriate pharmacokinetic (PK) modeling framework. Physiologically-Based Pharmacokinetic (PBPK) and Population PK (PopPK) modeling are two powerful, yet philosophically distinct, approaches. This analysis details their application notes and protocols, comparing their utility in the development of large molecule therapeutics.
Quantitative Comparison of PBPK vs. PopPK Approaches
The following table summarizes the core characteristics, data requirements, and applications of PBPK and PopPK models for therapeutic proteins.
Table 1: Core Comparison of PBPK and PopPK Modeling for Therapeutic Proteins
| Aspect | PBPK Modeling | PopPK Modeling |
|---|---|---|
| Theoretical Basis | Mechanism-driven; based on human physiology and drug-specific parameters. | Data-driven; uses mathematical functions to describe drug disposition in a population. |
| Structural Model | Defined by interconnected anatomical compartments (organs/tissues) with blood flow. | Defined by empirical compartments (central, peripheral) without physiological identity. |
| Key Inputs | Physiological parameters (organ volumes, blood flows), drug-specific parameters (e.g., target affinity, FcRn binding, lymph flow). | Observed concentration-time data from the target population. |
| Typical Outputs | Tissue concentration-time profiles, insights into mechanisms of distribution and clearance. | Population mean PK parameters, estimates of inter-individual variability (IIV), and covariate effects. |
| Primary Goal | Prediction & Understanding: Predict PK in untested scenarios (e.g., first-in-human, organ impairment) and understand tissue distribution. | Description & Estimation: Describe observed data variability and quantify impact of patient factors (e.g., weight, ADA). |
| Data Requirement | Primarily pre-clinical data (in vitro, in silico, animal studies). | Rich clinical PK data from Phase 1/2/3 trials. |
| Strength | Strong predictive capability for novel settings; integrates knowledge of biology and drug properties. | Efficiently handles sparse, real-world clinical data; identifies clinical covariates. |
| Weakness | Model complexity; requires extensive prior knowledge; may over-parameterize. | Limited extrapolation capability; provides less insight into mechanistic drivers. |
Application Notes
Application Note AN-01: PBPK for First-in-Human (FIH) Dose Prediction of a Novel mAb
- Objective: To predict human serum and target tissue (e.g., synovium for arthritis) PK for a novel anti-TNFα mAb using pre-clinical data, reducing uncertainty in FIH dose selection.
- Rationale: PBPK models leverage species-invariant physiology and scale drug-specific parameters (e.g., binding kinetics) from in vitro assays, enabling human PK prediction prior to clinical data generation.
- Protocol Link: See Protocol P-01.
Application Note AN-02: PopPK for Characterizing the Impact of Anti-Drug Antibodies (ADA) on Clearance
- Objective: To quantify the increase in clearance for a therapeutic enzyme in the presence of ADA and identify other clinical covariates (body size, disease severity) influencing exposure.
- Rationale: PopPK models are ideal for analyzing sparse Phase 3 data, where ADA status is measured periodically. Nonlinear mixed-effects modeling can statistically distinguish ADA-positive and ADA-negative subpopulations.
- Protocol Link: See Protocol P-02.
Experimental Protocols
Protocol P-01: Developing a Minimal PBPK (mPBPK) Model for a Monoclonal Antibody
Title: In Vitro to In Vivo Workflow for mAb PBPK Modeling.
1. Objectives: To construct and qualify a human mPBPK model for a mAb using pre-clinical data, enabling prediction of human serum and tissue PK. 2. Materials & Reagents: * Research Reagent Solutions: * Surface Plasmon Resonance (SPR) or Biolayer Interferometry (BLI) System: To measure antigen-binding affinity (KD, kon, koff). * FcRn Affinity Chromatography or SPR at pH 6.0 & 7.4: To quantify FcRn binding parameters critical for mAb half-life. * Recombinant Human Antigen: For target binding assays. * Animal Serum/Plasma: From PK studies in relevant species (e.g., mouse, monkey). * PBPK Software Platform: (e.g., GastroPlus, Simcyp, PK-Sim, or MATLAB/Python with differential equation solvers). 3. Procedure: 1. Data Collection: Assemble in vitro parameters (Step 1 above) and in vivo PK data from preclinical species. 2. Model Structuring: Build a mPBPK model typically comprising plasma, lymph, and two tissue compartments (e.g., "rich" and "lean") with mechanistic lymph flow. 3. Parameterization: * Fix physiological parameters (volumes, flows, FcRn expression) from literature. * Fit drug-specific parameters (e.g., nonspecific clearance, tissue permeability) to preclinical PK data using optimization algorithms. 4. Allometric Scaling & Translation: Scale fitted parameters (e.g., clearances) to human using established principles (e.g., fixed exponent scaling). 5. Qualification & Sensitivity Analysis: Perform virtual FIH simulations. Conduct sensitivity analysis on key uncertain parameters (e.g., lymph flow rate). 4. Data Analysis: Compare simulated human PK profiles (Cmax, AUC, half-life) to clinically observed data (if available) or published benchmarks for similar mAbs.
Protocol P-02: Conducting a PopPK Analysis for a Therapeutic Protein in Phase 3 Data
Title: PopPK Model Development Workflow.
1. Objectives: To develop a PopPK model describing the population typical profile and sources of variability (IIV, covariates) for a therapeutic protein using Phase 3 trial data. 2. Materials & Reagents: * Research Reagent Solutions: * Nonlinear Mixed-Effects Modeling Software: (e.g., NONMEM, Monolix, Phoenix NLME). * Clinical Database: Containing dependent variable (DV=concentration), independent variables (TIME, DOSE), and patient covariates (e.g., WEIGHT, AGE, ADA status, albumin). * Data Visualization & Processing Tools: (e.g., R with ggplot2, Python with Pandas/NumPy). * Model Diagnostic Tools: For generating Visual Predictive Checks (VPC), goodness-of-fit plots. 3. Procedure: 1. Data Preparation: Clean and format the clinical database. Conduct exploratory data analysis (e.g., concentration vs. time plots stratified by covariates). 2. Base Model Development: * Test different structural models (1-, 2-compartment with IV/SC absorption). * Estimate population typical parameters (CL, V, etc.) and IIV (e.g., ω² on CL). * Select residual error model (e.g., proportional, additive). 3. Covariate Model Development: * Test plausible covariate-parameter relationships (e.g., WEIGHT on CL and V using allometric scaling; ADA status as a categorical covariate on CL). * Use stepwise forward inclusion (p<0.05) and backward elimination (p<0.01) based on objective function value. 4. Model Evaluation: * Assess goodness-of-fit plots (obs vs. pred, CWRES vs. time). * Perform Visual Predictive Check (VPC) or Prediction-Corrected VPC (pcVPC) to evaluate model predictive performance. * Conduct bootstrap analysis to assess parameter uncertainty. 4. Data Analysis: Report final parameter estimates with precision. Quantify the clinical impact of key covariates (e.g., "ADA-positive status increases typical CL by 120%").
The Role of PBPK in Multi-Scale Systems Pharmacology Models
Physiologically Based Pharmacokinetic (PBPK) modeling is a critical pillar within the broader paradigm of Multi-Scale Systems Pharmacology (MSP) models. In the context of therapeutic proteins and monoclonal antibodies (mAbs), PBPK models provide the essential physiological and anatomical framework that bridges molecular-scale drug-target interactions to organism-level clinical outcomes. By quantitatively integrating drug-specific properties (e.g., FcRn affinity, target-mediated drug disposition) with system-specific physiology (e.g., tissue volumes, blood flows, FcRn expression, target antigen density), PBPK models enable the prediction of pharmacokinetics (PK) and pharmacodynamics (PD) across scales, populations, and disease states.
Quantitative Data Integration in PBPK-MSP Models
The predictive power of PBPK-informed MSP models relies on curated, high-quality quantitative data. The tables below summarize key parameters.
Table 1: Core Physiological Parameters for mAb PBPK Models
| Parameter | Typical Value (Human, 70kg) | Variability Source | Relevance to mAbs/Therapeutic Proteins |
|---|---|---|---|
| Plasma Volume | ~3 L | Body weight, sex | Distribution volume for central compartment |
| Interstitial Volume | ~12 L | Body weight, tissue composition | Major distribution space for mAbs |
| Lymph Flow Rate | ~0.2-0.3 L/h | Body weight, physiology | Key driver of convective mAb transport from interstitium |
| Plasma Clearance (IgG1) | ~0.2-0.4 L/day | FcRn affinity, immunogenicity | Linear clearance pathway |
| FcRn Concentration (Endothelium) | ~0.1-0.5 µM | Tissue type, genetic factors | Determines nonlinear salvage from degradation |
| Capillary Permeability (PS) | Tissue-specific (e.g., High: Liver; Low: Muscle) | Vascular pore size, surface area | Controls extravasation rate |
Table 2: Key Drug-Specific Parameters for Therapeutic Proteins
| Parameter | Typical Range | Experimental Method | Impact on PBPK-MSP Model |
|---|---|---|---|
| Target Affinity (KD) | pM to nM | Surface Plasmon Resonance (SPR) | Drives target-mediated drug disposition (TMDD) |
| Internalization Rate (kint) | 0.01-0.5 h⁻¹ | Cell-based assays with radiolabel/flow | Determines elimination via TMDD pathway |
| FcRn Affinity (KD at pH 6.0) | 0.1-5 µM | SPR at endosomal pH | Controls recycling and terminal half-life |
| Non-Specific Endocytosis (kns) | 0.001-0.01 h⁻¹ | In vitro cellular uptake studies | Contributes to linear clearance |
Application Notes
Integrating TMDD into a Full-PBPK Framework
For mAbs, TMDD is a small-scale (cellular/molecular) process that must be embedded within a whole-body PBPK model. The PBPK model defines the drug supply rate to the tissue compartment where the target is expressed. The local drug and target concentrations then drive the nonlinear binding, internalization, and degradation dynamics described by TMDD kinetics. This integration allows for predicting how changes in target expression (e.g., disease state, patient stratification) impact systemic PK and ultimately PD.
Translating from Preclinical to Clinical Scales
A primary role of PBPK in MSP is interspecies scaling. A mAb PBPK model parameterized with mouse or monkey physiology (organ weights, blood flows, FcRn expression) and drug parameters can be scaled to human by replacing the physiological platform with human parameters. Drug parameters (affinities, etc.) are typically assumed constant. This approach de-risks clinical entry by providing first-in-human PK predictions and guiding dose selection.
Simulating Pharmacodynamic Effects
In an MSP model, the PBPK module outputs time-course drug concentrations in plasma and various tissue interstitium. These concentrations serve as the input to a quantitative systems pharmacology (QSP) module describing the drug's mechanism of action (MoA). For an oncology mAb, this could involve linking tumor interstitial mAb concentration to receptor occupancy, downstream signaling inhibition, tumor cell apoptosis, and tumor growth dynamics.
Experimental Protocols
Protocol 1: Determination of mAb Tissue-Specific Lymphatic Drainage Rate
Objective: To experimentally estimate the rate constant of antibody movement from a tissue interstitium to lymph, a critical parameter for PBPK model fitting. Materials: Radiolabeled or fluorescently labeled mAb (e.g., 125I-IgG), microdialysis probe system, lymph cannula, animal model (rat). Procedure:
- Anesthetize and prepare the animal. Cannulate the efferent lymphatic duct draining the tissue of interest (e.g., hind leg).
- Inject a low, trace dose of the labeled mAb directly into the tissue interstitium.
- Collect lymph samples sequentially over 48-72 hours. Simultaneously, collect serial blood samples.
- Quantify radioactivity/fluorescence in all samples.
- Analyze data using a two-compartment model (tissue interstitial lymph) to estimate the lymphatic drainage rate constant (k_lymph).
- Scale the parameter using known allometric relationships for incorporation into the human PBPK model.
Protocol 2: In Vitro Estimation of Cellular Endocytosis and Degradation Rates
Objective: To quantify the rate constants for target-mediated internalization (kint) and degradation, necessary for TMDD-PBPK models. Materials: Target-positive cell line, mAb of interest, pH-sensitive fluorescent dye (e.g., pHrodo), flow cytometer, bioreactor for continuous collection. Procedure:
- Label the mAb with pHrodo dye according to manufacturer's protocol. This dye fluoresces intensely in acidic environments (e.g., endosomes, lysosomes).
- Incubate cells with the labeled mAb at 4°C to allow surface binding without internalization.
- Wash cells thoroughly and resuspend in fresh, pre-warmed medium. Immediately transfer to a bioreactor setup with a continuous micro-sampler.
- Initiate the experiment by raising the temperature to 37°C. Collect small supernatant aliquots continuously or at very frequent intervals (e.g., every 2-5 minutes) for 2-4 hours.
- Analyze supernatant fluorescence by plate reader. The increasing fluorescence in the supernatant over time corresponds to the degradation and release of fluorescent fragments from lysosomes.
- Fit the fluorescence-time data to a first-order degradation model to estimate the degradation rate constant. Use complementary flow cytometry data on cell-associated fluorescence decay to estimate the internalization rate constant (kint).
Visualization of Key Concepts
PBPK and TMDD Model Integration Pathway
Multi-Scale Systems Pharmacology Workflow
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for PBPK-MSP of Therapeutic Proteins
| Item | Function in PBPK-MSP Context | Example/Notes |
|---|---|---|
| pH-Sensitive Fluorescent Dyes (e.g., pHrodo) | Label antibodies to track internalization and degradation in live cells via fluorescence increase in acidic compartments. | Critical for estimating cellular rate constants (kint, kdeg) for TMDD models. |
| Biotinylation & Streptavidin-Based Capture Kits | For site-specific labeling of mAbs or creating surface-immobilized targets for kinetic binding studies. | Used in SPR or BLI to measure target and FcRn binding kinetics (kon, koff, KD). |
| Recombinant Human FcRn Protein | For in vitro binding assays at endosomal pH (6.0) to quantify the key recycling interaction. | Parameterizes the critical salvage mechanism in PBPK models. |
| Radiolabeled Isotopes (125I, 111In) | For precise, quantitative tissue distribution and pharmacokinetic studies in preclinical models. | Gold standard for generating data to calibrate tissue distribution parameters in PBPK models. |
| Microdialysis Systems | To sample free, unbound drug concentration in the interstitial fluid of specific tissues in vivo. | Provides direct data for validating predicted tissue interstitial concentrations from PBPK models. |
| QSP-Ready Cell Signaling Panels | Multiplex phosphoprotein or gene expression assays to quantify downstream pharmacological effects. | Generates data to parameterize and validate the PD/QSP module linked to the PBPK output. |
| PBPK Modeling Software | Platforms (e.g., GastroPlus, Simcyp, PK-Sim) with built-in mAb frameworks for model construction and simulation. | Essential tool for integrating data and executing MSP simulations. |
Benchmarking Against Emerging AI/ML Approaches in Pharmacokinetic Prediction
The integration of AI and ML into pharmacokinetic (PK) prediction represents a paradigm shift for PBPK modeling of monoclonal antibodies (mAbs) and therapeutic proteins. Traditional mechanistic PBPK models for large molecules rely on detailed physiological parameters, FcRn recycling, and target-mediated drug disposition. AI/ML approaches offer data-driven alternatives or hybrid enhancements that can accelerate development, improve prediction accuracy for novel modalities, and identify complex, non-linear relationships not easily captured by classic models. This document provides application notes and protocols for systematically benchmarking these emerging approaches against established PBPK frameworks in the context of large molecule drug development.
The table below summarizes recent (2022-2024) key AI/ML methodologies applied to PK prediction for proteins, with a focus on performance metrics relative to traditional PBPK.
Table 1: Benchmarking of AI/ML Approaches for PK Prediction of mAbs/Therapeutic Proteins
| Approach Category | Key Algorithm/Model | Reported Application (Dataset Size) | Performance Metric vs. Traditional PBPK | Key Advantage | Primary Limitation |
|---|---|---|---|---|---|
| Pure ML Predictive | Gradient Boosting (XGBoost/LightGBM) | Human mAb PK (n=~50 compounds) | RMSE reduced by 25-40% for CL and Vss predictions in early development | Handles sparse, heterogeneous data; rapid screening | Low mechanistic insight; poor extrapolation |
| Deep Learning (NN) | 1D-CNN or Graph Neural Networks (GNN) | Peptide & mAb SC absorption (n=~100 studies) | AUC prediction accuracy improved by 15-20% for complex SC profiles | Learns from raw sequence/structure data | High data requirement; "black box" nature |
| Hybrid PBPK-ML | ML-informed PBPK parameters (e.g., ML-predicted CL) | PBPK for bispecific antibodies (n=~20) | Reduced PBPK model calibration time by >50% | Balances mechanism and data-driven learning | Integration complexity; dependency on PBPK platform |
| Physicochemical ML | Random Forest & SHAP analysis | Predicting mAb tissue:plasma ratios (n=~30 mAbs) | Outperformed traditional QSPR models (R² >0.8 vs. <0.6) | Provides feature importance (interpretability) | Limited to chemical space of training data |
| Time-Series ML | Long Short-Term Memory (LSTM) Networks | Predicting patient-level PK variability (n=~500 patients) | Captured variability 30% more accurately than population PBPK | Excellent for sequential/time-dependent data | Requires rich, longitudinal patient data |
Experimental Protocols for Benchmarking
Protocol 1: Benchmarking a Hybrid PBPK-ML Model for mAB Clearance Prediction
Objective: To compare the predictive performance of a Hybrid PBPK-ML model against a full PBPK model and a standalone ML model for predicting human clearance of mAbs.
Materials (Research Reagent Solutions):
- Dataset: Curated proprietary or public dataset of mAbs with reported human PK parameters (CL, Vss, t1/2). Minimum n=40 unique molecules.
- Software: PBPK platform (e.g., Simbiology, PK-Sim, GastroPlus), Python/R with ML libraries (scikit-learn, XGBoost, TensorFlow/PyTorch).
- Features: Molecular descriptors (pI, MW, hydrophobicity index), in vitro assay data (FcRn affinity at pH 6.0 & 7.4, nonspecific binding), in silico developability scores.
Procedure:
- Data Curation: Assemble a dataset. Perform train/validation/test split (e.g., 70/15/15).
- Baseline PBPK Model: Develop a minimal PBPK model (2-4 tissue compartments, FcRn recycling). Calibrate using in vitro inputs for each mAb in the training set.
- Standalone ML Model: Train a Gradient Boosting Regressor (e.g., XGBoost) on the training set to predict human CL directly from molecular/ in vitro features.
- Hybrid Model Construction:
- Use the trained ML model from step 3 to predict the CL input parameter for the PBPK model.
- Keep all other PBPK parameters (e.g., Vss, tissue rates) mechanistically derived.
- Run the PBPK simulation with the ML-predicted CL.
- Benchmarking: On the held-out test set, compare predicted vs. observed serum concentration-time profiles. Use metrics: Root Mean Square Error (RMSE), Mean Absolute Error (MAE), and AUC ratio (predicted/observed).
- Analysis: Perform a statistical comparison (e.g., paired t-test) of error metrics across the three approaches.
Protocol 2: Evaluating Deep Learning for Subcutaneous Absorption Prediction
Objective: To assess the ability of a Convolutional Neural Network (CNN) to predict the absorption rate constant (ka) and bioavailability (F) of therapeutic proteins after subcutaneous (SC) administration compared to a classical SC absorption PBPK model.
Materials (Research Reagent Solutions):
- Dataset: Time-series plasma concentration data from SC administration studies (multiple species). Include formulation details (concentration, viscosity).
- Software: Python with deep learning stack (TensorFlow/Keras), data preprocessing tools.
- Input Features: Amino acid sequence (one-hot encoded), formulation properties, dose, injection volume.
Procedure:
- Data Preprocessing: Standardize PK data. Encode protein sequences into 2D arrays (sequence length x amino acid features).
- Model Architecture: Design a 1D-CNN with layers for sequence feature extraction, followed by dense layers for processing formulation/dose data, merging into a final regression layer for ka and F.
- Training: Train the CNN on the training dataset. Use k-fold cross-validation.
- PBPK Baseline: Develop a standard SC absorption PBPK model (lymphatic flow, pre-systemic catabolism).
- Validation: Predict SC profiles for a test set of novel constructs. Compare CNN predictions vs. PBPK model simulations against actual data using metrics of profile similarity (e.g., Visual Predictive Check, RMSE of entire profile).
Visualizations: Workflows and Relationships
Diagram 1: Benchmarking Workflow for PK Prediction Models
Diagram 2: Information Flow in a Hybrid PBPK-ML Model
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials and Tools for AI/ML-PBPK Benchmarking
| Item / Solution | Function in Benchmarking | Example/Provider |
|---|---|---|
| Curated PK Database | Gold-standard dataset for training and testing models; must include molecule attributes, in vitro data, and in vivo PK. | Proprietary company database, public sources (e.g., PubMed, DrugBank). |
| PBPK Software Platform | Provides the mechanistic modeling framework against which AI/ML is benchmarked. | Simbiology (MATLAB), PK-Sim (Open Systems Pharmacology), GastroPlus. |
| ML Development Environment | Toolkit for building, training, and validating pure ML and hybrid models. | Python (scikit-learn, XGBoost, PyTorch), R (tidymodels, caret). |
| In Silico Developability Suite | Generates molecular descriptors and predicted liabilities (aggregation, hydrophobicity) as ML model inputs. | Schrödinger Biologics, MOE, custom sequence-based pipelines. |
| In Vitro FcRn Affinity Assay Kit | Provides critical in vitro parameter for both traditional PBPK (binding rate) and ML feature sets. | Surface Plasmon Resonance (SPR) based kits (e.g., Cytiva). |
| High-Performance Computing (HPC) | Enables rapid hyperparameter tuning for ML models and large-scale PBPK simulations. | Cloud platforms (AWS, GCP), local computing clusters. |
| Data Visualization & Stats Package | For creating standardized comparison plots (observed vs. predicted) and statistical testing. | R (ggplot2), Python (Matplotlib, Seaborn), GraphPad Prism. |
Conclusion
PBPK modeling for monoclonal antibodies and therapeutic proteins has matured into an indispensable tool in modern drug development, offering a mechanistic framework to navigate the unique complexities of large molecule pharmacokinetics. From foundational principles to advanced applications, this approach enables more informed decision-making in dose selection, scaling, and predicting behavior in special populations. While challenges in parameterization and validation persist, ongoing integration with quantitative systems pharmacology (QSP) and machine learning heralds a future of increasingly predictive and personalized models. For researchers and drug developers, mastering biologics PBPK is no longer optional but a critical competency for accelerating the development of safer and more effective protein-based therapeutics, ultimately streamlining the path from bench to bedside and strengthening regulatory submissions.