Research Articles
Mastering the Cheng-Prusoff Equation: A Practical Guide for Accurate Ki Determination in Binding Assays
This comprehensive guide demystifies the Cheng-Prusoff equation, a cornerstone of receptor-ligand binding analysis.
Mastering the CellTiter-Glo Assay: A Complete Guide to Accurate Viability Measurement in Drug Discovery
This comprehensive guide details the complete CellTiter-Glo Luminescent Cell Viability Assay protocol for researchers and drug development professionals.
CRISPR Revolution: Accelerating and Refining Target Validation in Drug Discovery
This article provides a comprehensive guide for researchers and drug development professionals on the transformative role of CRISPR-Cas technology in target validation.
From Lab to Bedside: A Comprehensive Guide to Modern CAR-T Cell Manufacturing and Clinical Integration
This article provides a detailed, current overview of the end-to-end process of CAR-T cell therapy, tailored for researchers, scientists, and drug development professionals.
Beyond the Class: How the BCS Framework Drives Modern Drug Discovery & Development
This article provides a comprehensive exploration of the Biopharmaceutics Classification System (BCS) as a critical, predictive tool in contemporary drug discovery and development.
Bayesian Optimal Design for Dose-Response Studies: Maximizing Efficiency in Drug Development
This article provides a comprehensive guide to Bayesian optimal design (BOD) for dose-response modeling, targeted at researchers and professionals in pharmaceutical development.
Bayesian Biomarker Discovery: A Framework for Precision Pharmacodynamic Analysis in Drug Development
This article provides a comprehensive guide to Bayesian frameworks for identifying pharmacodynamic (PD) biomarkers, which are critical for understanding drug mechanism of action and predicting clinical response.
BCS Class II vs. Class IV Drug Development: Formulation Strategies for Poor Solubility and Permeability
This article provides a comprehensive analysis of formulation strategies for Biopharmaceutics Classification System (BCS) Class II (low solubility, high permeability) and Class IV (low solubility, low permeability) drugs.
Overcoming Solubility Barriers: Advanced Strategies for BCS Class II and IV Drug Development
This article provides a comprehensive review of the solubility challenges inherent to Biopharmaceutics Classification System (BCS) Class II (low solubility, high permeability) and Class IV (low solubility, low permeability) drugs.
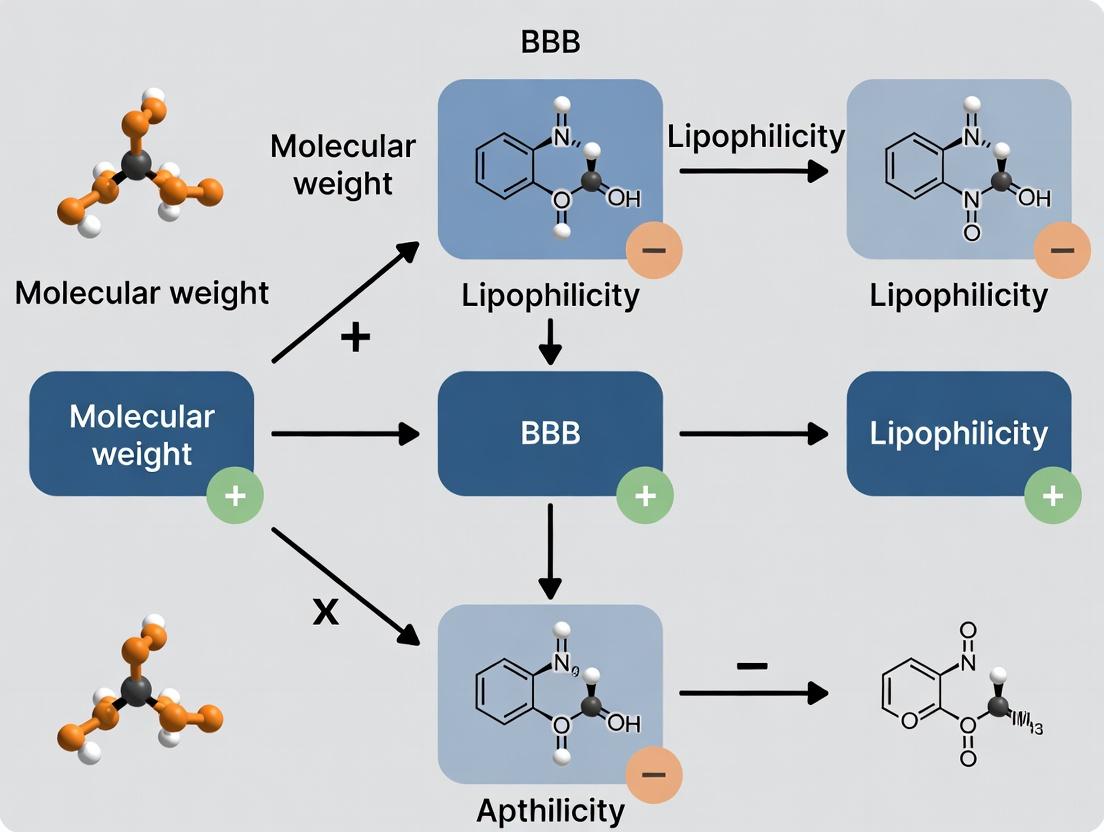
Crossing the Barrier: Molecular Weight, Lipophilicity, and BBB Permeability in CNS Drug Development
This article provides a comprehensive analysis of the two primary physicochemical determinants of Blood-Brain Barrier (BBB) permeability: molecular weight (MW) and lipophilicity (often measured as LogP/LogD).