Research Articles
ICH Stability Testing Guidelines Q1A(R2) to Q1E: A Comprehensive Guide for Drug Substance Shelf Life
This detailed guide provides drug development professionals with a current, in-depth analysis of ICH stability testing guidelines for new drug substances.
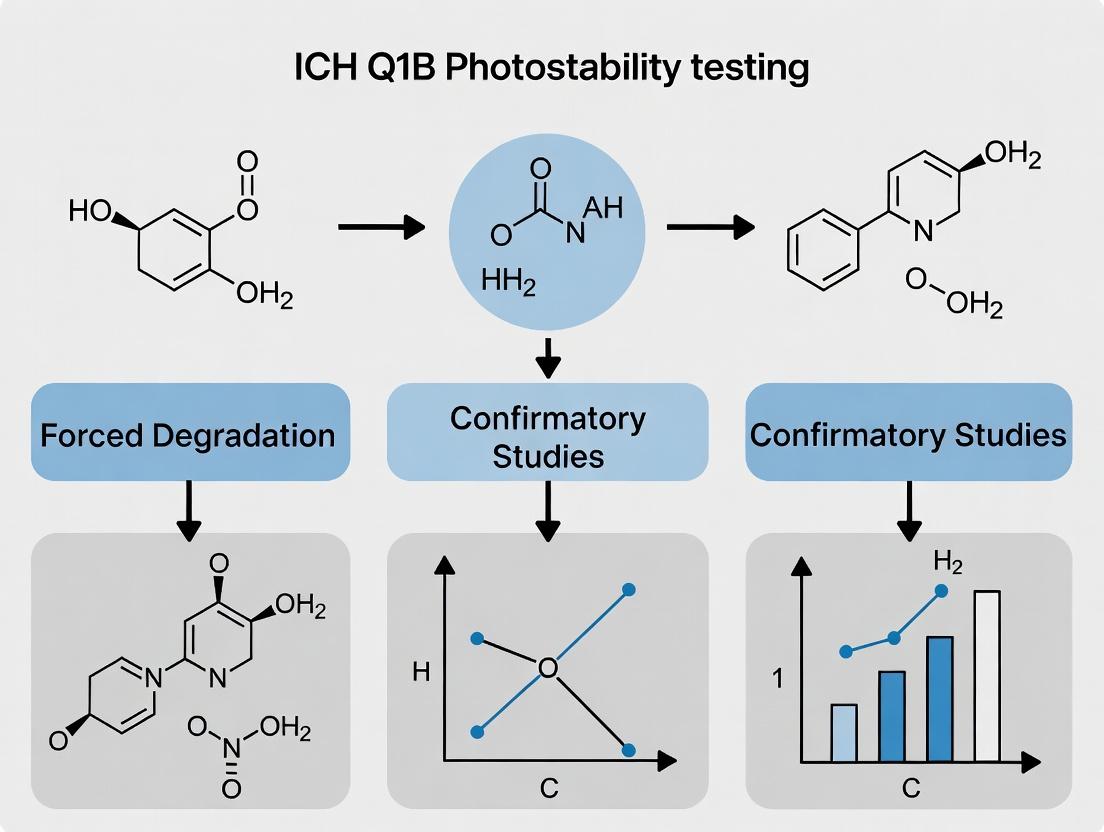
ICH Q1B Photostability Testing Explained: Strategic Application of Forced Degradation vs. Confirmatory Studies in Drug Development
This article provides a comprehensive guide to ICH Q1B photostability testing for drug development professionals, differentiating between forced degradation (stress) and confirmatory studies.
ICH Q1A(R2) Stability Testing Demystified: Your Complete Guide to Registration Data Packages
This comprehensive guide provides drug development professionals with a detailed breakdown of the ICH Q1A(R2) stability data package requirements for drug registration.
ICH Q1 Stability Testing Guidelines 2025: A Comprehensive Analysis of the Draft Revision and Its Impact on Drug Development
This article provides a detailed overview of the proposed ICH Q1A(R3)/Q1B(R2)/Q1C(R2) 2025 draft guidelines for stability testing of new drug substances and products.
HTS Hit Confirmation and Counter-Screening: A Strategic Guide for Robust Drug Discovery
This comprehensive guide outlines critical strategies for confirming hits from high-throughput screening (HTS) campaigns while mitigating false positives.
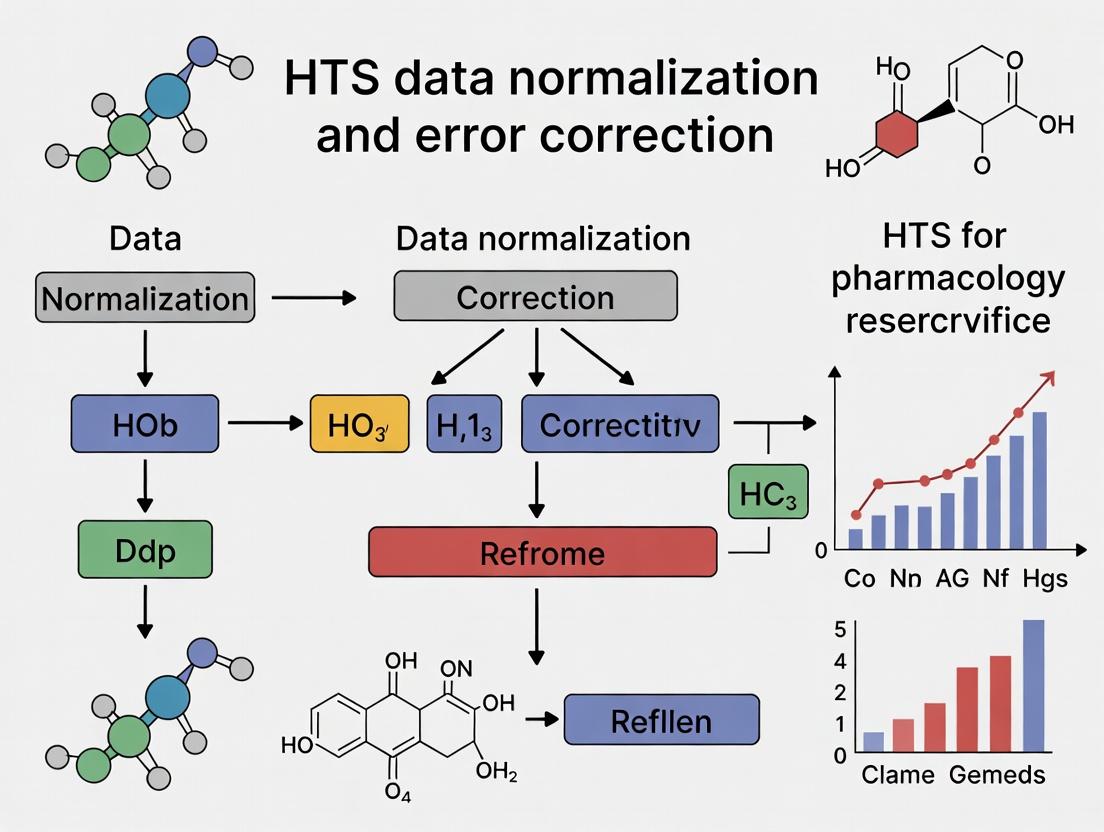
From Noise to Knowledge: A Modern Guide to HTS Data Normalization and Error Correction
This comprehensive guide for researchers and drug development professionals demystifies the critical steps of normalization and error correction in High-Throughput Screening (HTS).
Advanced HTRF Assays for Cannabinoid Receptor Drug Discovery: Validation, Protocols & Optimization
This comprehensive guide details the application of HTRF® (Homogeneous Time-Resolved Fluorescence) technology for high-throughput screening and mechanistic studies of cannabinoid receptors CB1R and CB2R.
HPLC vs UPLC: Choosing the Optimal Chromatography for Complex Drug Mixture Resolution in Modern Pharma
This comprehensive guide explores the critical decision between High-Performance Liquid Chromatography (HPLC) and Ultra-Performance Liquid Chromatography (UPLC) for resolving complex drug mixtures in pharmaceutical research and development.
Advanced HPLC Techniques for Therapeutic Drug Monitoring of Anticonvulsants: Method Development, Validation, and Clinical Application
This comprehensive article addresses the critical role of High-Performance Liquid Chromatography (HPLC) in the therapeutic drug monitoring (TDM) of anticonvulsant medications.
Sharpening Your Peaks: A Comprehensive Guide to Diagnosing and Fixing HPLC/UPLC Peak Broadening
This definitive guide for chromatographers addresses the critical challenge of peak broadening in HPLC and UPLC.